Last updated on Oct 23, 2015
Get the free ACRIN Adverse Event Log
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Adverse Event Log
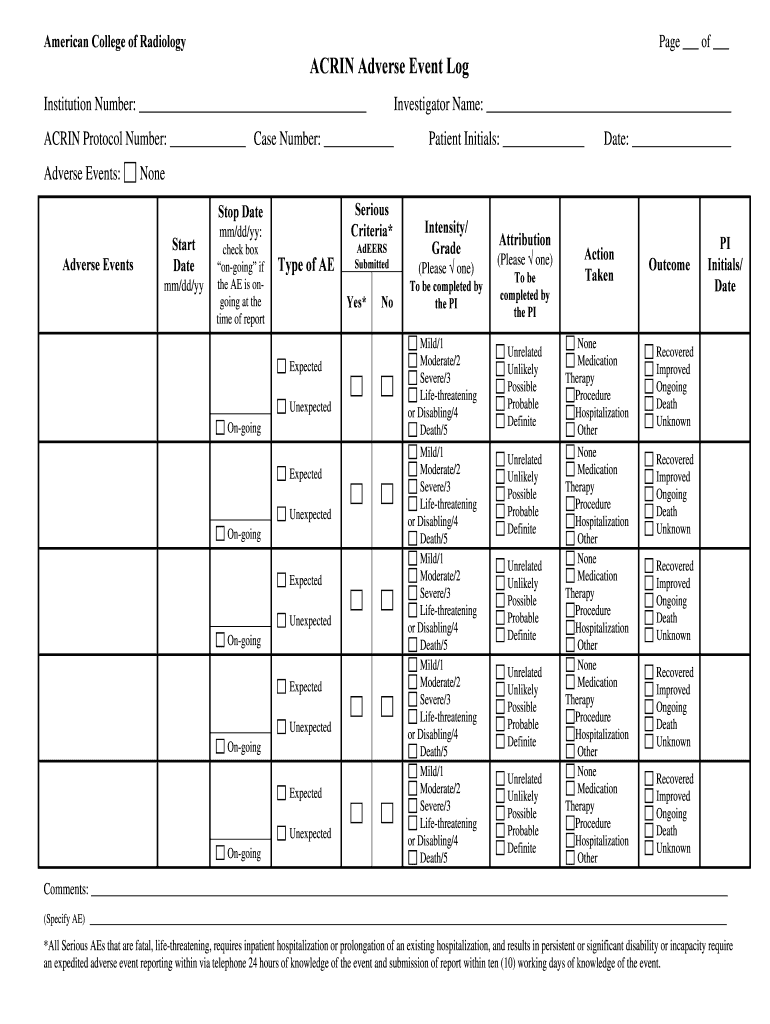
The ACRIN Adverse Event Log is a healthcare form used by investigators to document and report adverse events in clinical trials.
pdfFiller scores top ratings on review platforms




Who needs Adverse Event Log?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Adverse Event Log
What is the ACRIN Adverse Event Log?
The ACRIN Adverse Event Log is a crucial tool used to document and report adverse events during clinical trials. This log plays a significant role in ensuring patient safety by providing a systematic approach to tracking these events. Its importance extends to regulatory compliance, as accurate documentation is essential for adhering to the standards set by governing bodies. This form is used by investigators to capture details associated with adverse events, ultimately safeguarding participants' health in clinical research.
Purpose and Benefits of the ACRIN Adverse Event Log
The ACRIN Adverse Event Log serves multiple purposes within clinical trials, primarily focusing on the effective tracking of adverse events. Timely and accurate entries in this log enhance the quality of clinical research by identifying potential safety issues early on. Benefits also include the improvement of study protocols and the enhancement of patient care based on the feedback collected in the log.
Key Features of the ACRIN Adverse Event Log
This log encompasses several key features that facilitate comprehensive adverse event reporting. The essential sections of the form include:
-
Patient initials
-
Protocol number
-
Type of event
-
Actions taken
Each section is designed to gather critical information, including specific reporting requirements for serious adverse events, ensuring that investigators meet their obligations during clinical trials.
Who Needs to Use the ACRIN Adverse Event Log?
Primarily, the ACRIN Adverse Event Log is intended for investigators who are responsible for monitoring patient safety throughout clinical trials. Their commitment to utilizing this log is crucial, as they must accurately document all relevant adverse events. Institutions involved in clinical trials also rely on this log, as it supports research teams in maintaining compliance and enhancing overall research integrity.
How to Fill Out the ACRIN Adverse Event Log Online
Completing the ACRIN Adverse Event Log online involves several straightforward steps:
-
Begin with entering patient initials.
-
Fill in the protocol number.
-
Select the type of adverse event from the provided options.
-
Document the intensity and attribution of the event.
-
Detail actions taken in response to the event.
Ensure that all entries are accurate and complete to promote effective reporting and monitoring of adverse events.
Common Errors to Avoid When Completing the ACRIN Adverse Event Log
While filling out the ACRIN Adverse Event Log, users should be vigilant to avoid typical errors that could compromise the document's integrity. Common mistakes include:
-
Omitting required signatures
-
Entering incorrect patient information
Double-checking all entries before submission is crucial to maintaining the log's accuracy.
Submission Process for the ACRIN Adverse Event Log
Submitting the completed ACRIN Adverse Event Log can be done electronically or via hard copies. The submission should include the following requirements:
-
Electronic submissions must follow the procedure outlined in the platform you are using.
-
Hard copies should be sent to the designated address specified for submissions.
Keeping a record of submissions and obtaining confirmation receipts can help in tracking the status of the log once filed.
Security and Compliance of the ACRIN Adverse Event Log
Data protection and confidentiality are paramount in handling the ACRIN Adverse Event Log. Various security measures are enforced to protect sensitive information, ensuring compliance with regulations such as HIPAA and GDPR. These measures guarantee that the data remains secure while still being accessible for necessary reporting purposes.
Why Choose pdfFiller to Complete the ACRIN Adverse Event Log?
pdfFiller significantly enhances the process of completing the ACRIN Adverse Event Log by offering a cloud-based platform. Users benefit from features that streamline filling and eSigning, including:
-
No downloads required for access
-
Intuitive user interface
-
Robust security measures for document handling
These capabilities make pdfFiller a reliable choice for completing healthcare forms efficiently and securely.
Visual Example of a Completed ACRIN Adverse Event Log
For reference, a visual example of a completed ACRIN Adverse Event Log can be incredibly helpful for users. This should include annotations explaining each section, aiding in the interpretation of various fields. Understanding how to effectively fill in the log will promote better compliance and accurate reporting.
How to fill out the Adverse Event Log
-
1.Access pdfFiller and locate the ACRIN Adverse Event Log form in the healthcare forms section. Open the form by clicking on it and selecting 'Edit' to begin your entries.
-
2.Familiarize yourself with the form layout, noting the sections for patient data and adverse event details. Utilize pdfFiller's tools to navigate smoothly between fields.
-
3.Before entering information, gather all necessary data such as patient initials, protocol number, details of the adverse event, intensity, attribution, and any actions taken regarding the event.
-
4.Start by entering patient initials in the designated field, followed by the protocol number to ensure accurate tracking of the subject's data.
-
5.Proceed to the adverse event section, filling in details such as the type of adverse event and its intensity, tapping into the guidelines provided within the form for clarity.
-
6.Utilize checkboxes if applicable and ensure you attribute the event accurately as per the instructions provided. These details are crucial for compliance.
-
7.Review the completed form thoroughly, checking for any blank fields and ensuring all information is clear and legible before finalization.
-
8.Once satisfied with your entries, save your work on pdfFiller. You may choose to download a PDF version of the completed form for easy distribution or submission.
-
9.Finally, submit your completed ACRIN Adverse Event Log according to your organization’s protocols, whether electronically or via hard copy, ensuring it reaches the relevant authority promptly.

Who is eligible to use the ACRIN Adverse Event Log?
Any investigator involved in clinical trials is eligible to use the ACRIN Adverse Event Log for documenting adverse events and ensuring timely reporting.
What information do I need before completing the form?
Before starting, gather patient initials, protocol number, details of the adverse event, severity, attribution, and actions taken to ensure thorough and accurate reporting.
How do I submit the completed ACRIN Adverse Event Log?
Once completed, you can save the form on pdfFiller, then download it for submission or follow your organization’s specified submission procedure.
Are there deadlines for submitting the ACRIN Adverse Event Log?
It's crucial to submit the form promptly, especially for serious adverse events that require expedited reporting as per regulatory requirements.
What are common mistakes to avoid while filling out this form?
Common mistakes include leaving fields blank, providing incomplete information on adverse event details, or not signing where required by the investigator.
Is notarization required for the ACRIN Adverse Event Log?
No, notarization is not required for the ACRIN Adverse Event Log, simplifying the process for investigators.
How long does it take to process the ACRIN Adverse Event Log?
Processing times can vary based on the organization’s protocols, but timely submission is essential to ensure appropriate follow-up and action.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















