Last updated on Oct 24, 2015

Get the free Human Participants Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Human Participants Form
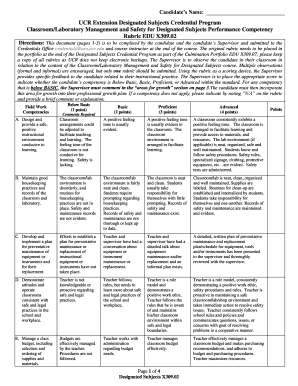
The Human Participants Form is a critical document used by student researchers to seek approval for research involving human subjects outside regulated institutions.
pdfFiller scores top ratings on review platforms




Who needs Human Participants Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Human Participants Form
What is the Human Participants Form?
The Human Participants Form is a vital document utilized in research involving human subjects. It plays a critical role in ensuring that ethical standards are upheld throughout the research process. This form must be completed by student researchers in collaboration with their adult sponsors, requiring institutional review board (IRB) approval to safeguard participants.
Completing the Human Participants Form is mandatory for those involved in research that touches on human subjects. This process not only identifies potential risks but also establishes the framework for ethical research practices.
Purpose and Benefits of the Human Participants Form
The primary goal of the Human Participants Form is to promote ethical research practices. By utilizing this form, researchers ensure that they adhere to necessary ethical standards within their studies. This form benefits student researchers and adult sponsors by clarifying the protocols needed for safe and compliant research practices.
-
Supports ethical standards related to human subjects research
-
Enhances compliance for student researchers and sponsors
-
Ensures safety assessments are conducted
Who Needs the Human Participants Form?
The following roles are essential players in the completion of the Human Participants Form:
-
Student Researchers
-
Adult Sponsors
-
Medical or Mental Health Professionals
-
School Administrators
-
Educators
Each role carries specific responsibilities, particularly regarding the signature requirements necessary for completing the form and ensuring that all stakeholders are involved in the process.
Key Features of the Human Participants Form
This form is comprehensive in its structure, consisting of several key sections that facilitate research submission and oversight. Key elements include:
-
Research plan submission
-
Informed consent documentation
-
IRB approval criteria
-
Minimal risk research assessments
-
Signature collection requirements
Each section is crafted to capture essential information while maintaining clarity and functionality for users in diverse research environments.
How to Fill Out the Human Participants Form Online (Step-by-Step)
To efficiently fill out the Human Participants Form online using pdfFiller, follow the steps below:
-
Access the Human Participants Form template on pdfFiller.
-
Complete all required fields with accurate information.
-
Utilize the eSigning feature for necessary signatures.
-
Review the completed form for accuracy.
-
Save the document securely within the pdfFiller platform.
This guide ensures that every user can navigate the process with ease, enhancing the overall experience for those completing the form.
Common Errors and How to Avoid Them
While filling out the Human Participants Form, various mistakes can hinder the process. Common errors include:
-
Incomplete sections or missing signatures
-
Incorrect or unclear information
-
Failure to adhere to deadlines
To avoid these pitfalls, it is critical to review the form comprehensively before submission. Ensuring accuracy and completeness can significantly reduce the chances of delays or rejections.
Submission Methods and Deadlines for the Human Participants Form
Submitting the completed Human Participants Form is crucial for gaining the necessary IRB approval. Follow these methods for submission:
-
Email submission to the designated IRB address
-
Physical submission to the appropriate research office
Pay attention to the submission deadlines, which are particularly vital for science fair projects and similar research initiatives. Missing these deadlines could impede the research timeline.
What Happens After You Submit the Human Participants Form?
After submission, the Human Participants Form undergoes a thorough review process by the IRB. Stakeholders should remain informed about the following aspects:
-
Anticipated confirmation timelines from the IRB
-
Methods for tracking the status of the form
-
Common reasons for potential rejection
This clarity allows researchers to understand what to expect in the aftermath of their submission, enhancing their preparedness for next steps.
Security and Compliance When Using the Human Participants Form
Data protection and compliance are paramount when utilizing the Human Participants Form. pdfFiller employs robust security measures, including:
-
256-bit encryption for data protection
-
Adherence to HIPAA and GDPR compliance standards
Ensuring confidentiality in research-related documents is essential, as it upholds the integrity of sensitive information shared throughout the research process.
Start Your Journey with the Human Participants Form Today!
Explore the ease and efficiency of filling out the Human Participants Form online with pdfFiller. By leveraging pdfFiller's tools, you can streamline the form completion process, enhance accuracy, and manage your documents securely. Taking advantage of these resources enables a smoother journey through research documentation.
How to fill out the Human Participants Form
-
1.Access the Human Participants Form by visiting the pdfFiller website and searching for 'Human Participants Form'. Ensure you're logged into your account before trying to locate the form.
-
2.Once you find the form, click on it to open it in the pdfFiller interface. Familiarize yourself with the layout and available tools for editing.
-
3.Before you begin filling out the form, gather all necessary information, including participant details, project objectives, and consent statements from your adult sponsor or supervisor.
-
4.Start filling in the required fields, using the text fields in the pdfFiller interface. Focus on entering clear and legible information concerning your research objectives and participant protocols.
-
5.Use checkboxes to indicate affirmations, ensuring that all required signatures are marked. Follow the prompts and guidance within pdfFiller to assist in completing any complex sections.
-
6.Review the form thoroughly after filling it out. Check for errors, confirming all sections are complete and signed by required parties to maintain compliance with IRB guidelines.
-
7.Once finalized, save your work by clicking the 'Save' button. You can also download the completed form in various formats or submit it directly to your institution from pdfFiller.

Who is eligible to fill out the Human Participants Form?
Eligibility includes student researchers conducting studies involving human participants, their adult sponsors, and qualified professionals like educators or medical experts who endorse the research.
What is the deadline for submitting the Human Participants Form?
Deadlines vary by institution and project type. It's crucial to check with your school or IRB for specific submission timelines relevant to your research project, especially for science fair competitions.
How do I submit the completed Human Participants Form?
After filling out the form, you can submit it to your institution electronically through pdfFiller or print it out for physical submission, depending on your institution's guidelines.
What supporting documents are required when submitting the form?
Typically, you must attach informed consent forms from participants and approval letters from adult sponsors. Additional documents may include research plans or ethical reviews.
What common mistakes should I avoid when completing the form?
Ensure all required signatures are obtained and check that no fields are left blank. Pay close attention to the IRB compliance requirements and participant consent specifics to avoid delays.
How long does it take for the Human Participants Form to be processed?
Processing times vary based on the institution's review schedule. Typically, allow several weeks for the processing of research forms, especially if an IRB review is needed.
What if I need to make changes after submitting the Human Participants Form?
If changes are necessary post-submission, contact your institution or IRB to understand their procedures for amendments. Prompt communication is essential to ensure compliance.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.