Last updated on Oct 25, 2015

Get the free IRB Reviewer Checklist Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

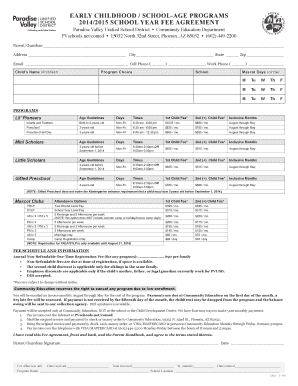
What is IRB Checklist
The IRB Reviewer Checklist Form is a fillable document used by Institutional Review Boards to evaluate research protocols effectively.
pdfFiller scores top ratings on review platforms




Who needs IRB Checklist?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to IRB Checklist
What is the IRB Reviewer Checklist Form?
The IRB Reviewer Checklist is a vital tool utilized by Institutional Review Boards (IRBs) to evaluate research protocols effectively. This document serves as a foundation for ensuring that research adheres to the highest ethical standards. By following a structured approach to protocol review, it helps maintain the integrity of clinical studies and supports the objectives of ethical research standards.
-
Defines the role of the checklist in assessing research protocols.
-
Highlights its significance in ethical evaluations by IRBs.
-
Makes connections to relevant concepts like clinical study and research ethics checklists.
Purpose and Benefits of the IRB Reviewer Checklist Form
This checklist is crucial for ensuring a thorough evaluation of all study documents, including the cover sheets and consent forms. By utilizing this form, researchers can support compliance with both regulatory requirements and ethical research standards.
-
Facilitates a comprehensive assessment of documentation.
-
Upholds compliance with regulatory guidelines.
-
Promotes adherence to ethical standards throughout research processes.
Key Features of the IRB Reviewer Checklist Form
The IRB Reviewer Checklist Form includes numerous essential features that simplify the review process. With a fillable form template, reviewers can provide detailed input using checkboxes designed for ease of use.
-
Sections for thorough protocol review and consent form evaluation.
-
Includes a study document evaluation section to ensure completeness.
-
Designed as an IRB form template to streamline the assessment process.
Who Needs the IRB Reviewer Checklist Form?
This form is primarily targeted at researchers and members of Institutional Review Boards. Both parties play a critical role in ensuring that ethical standards are maintained throughout the research process.
-
Researchers requiring detailed guidance on protocol evaluations.
-
Institutional Review Boards conducting comprehensive assessments for compliance.
How to Fill Out the IRB Reviewer Checklist Form Online (Step-by-Step)
Filling out the IRB Reviewer Checklist Form online is straightforward. Follow these step-by-step instructions for optimal completion:
-
Access the checklist from pdfFiller.
-
Fill in the required fields clearly and accurately.
-
Utilize checkboxes for each section as applicable.
-
Review your entries for completion, ensuring no fields are left blank.
-
Submit the form electronically after final review.
Common Errors and How to Avoid Them
It is essential to recognize and address common mistakes made while completing the IRB Reviewer Checklist Form. By being aware of these errors, users can mitigate issues before submission.
-
Omitting important sections or fields in the form.
-
Providing unclear information that may lead to misunderstandings.
-
Neglecting to double-check required documentation and guidelines.
Submission Methods and Delivery of the IRB Reviewer Checklist Form
Once completed, there are multiple methods for submitting the IRB Reviewer Checklist Form. Proper understanding of these methods will ensure compliance with institutional requirements.
-
Electronic submission via dedicated portals.
-
Print-out requirements for physical submission.
-
Awareness of state-specific regulations related to submission processes.
What Happens After You Submit the IRB Reviewer Checklist Form?
After submission, users should expect a review process that may require tracking of submission status. Understanding this phase can help manage expectations regarding feedback and outcomes.
-
Tracking submission status through designated platforms.
-
Receiving feedback within specified timeframes.
-
Recognizing potential rejection reasons and addressing corrective measures.
Security and Compliance for the IRB Reviewer Checklist Form
Users can rest assured about the security of their submissions while using pdfFiller. This platform adheres to strict compliance regulations to protect sensitive data.
-
256-bit encryption to safeguard information.
-
Full adherence to HIPAA and GDPR guidelines.
-
Regular audits to maintain compliance with security measures.
Empower Your Research with pdfFiller
Utilizing pdfFiller allows users to enhance their experience while filling out the IRB Reviewer Checklist Form. The platform offers user-friendly tools that streamline the entire form-filling process.
-
Cloud-based solutions for easy access and management.
-
User-oriented design that simplifies form completion.
-
Encouragement to start filling out the IRB Reviewer Checklist Form today.
How to fill out the IRB Checklist
-
1.To access the IRB Reviewer Checklist Form, visit pdfFiller's website and search for the form using its name in the search bar.
-
2.Once located, click on the form to open it. Ensure you are signed in or create an account if necessary.
-
3.Before filling out the form, gather all required documents including the research protocol, consent forms, and any other study materials relevant to the review.
-
4.Use the pdfFiller interface to navigate through the form fields. Click on each checkbox and text input area that needs filling.
-
5.Refer to the research documents you have gathered as you fill in the checklist, ensuring that you complete each section accurately.
-
6.After completing the form, review your entries for completeness and accuracy, checking that all required boxes are checked, and that all fields are filled out correctly.
-
7.Once satisfied with the form, you can save your progress or finalize the document in pdfFiller.
-
8.To save or download the completed IRB Reviewer Checklist Form, look for the download button or choose to share it directly from pdfFiller for submission.

Who is eligible to use the IRB Reviewer Checklist Form?
The IRB Reviewer Checklist Form is intended for use by Institutional Review Board members, clinical researchers, and administrators responsible for evaluating research protocols.
Are there specific deadlines for submitting this form?
Deadlines for submitting the IRB Reviewer Checklist Form may vary depending on the institution's policies. Check with your IRB office for specific submission timelines.
How do I submit the completed IRB Reviewer Checklist Form?
Completed forms can be submitted electronically through the institutional review board's submission portal or directly via email, depending on your institution's submission procedures.
What supporting documents are required with the IRB Reviewer Checklist?
It's often necessary to include related documents such as the study protocol, consent forms, and any supplementary materials required for the review process alongside the checklist.
What are common mistakes to avoid when filling out this form?
Common mistakes include skipping sections, failing to provide adequate explanations in blank fields, and not verifying compliance with ethical standards outlined in the protocol review.
How long does it take to process the IRB Reviewer Checklist Form?
Processing times vary but typically range from a few days to several weeks, depending on the complexity of the study and the IRB's workload. Confirm with your board for specific timelines.
Can I edit the form after submission?
Editing the IRB Reviewer Checklist Form after submission may depend on the institution's policies. It's advised to check with the IRB office about their procedures for amendment requests.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.