Last updated on Oct 30, 2015
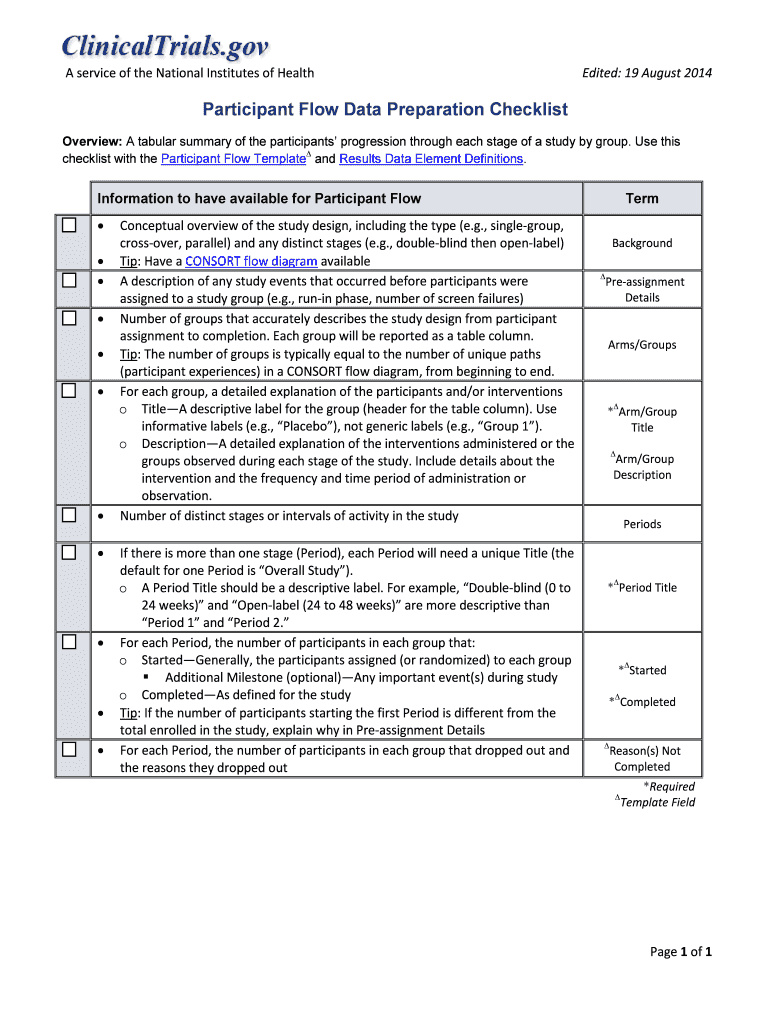
Get the free Participant Flow Data Preparation Checklist
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Flow Data Checklist
The Participant Flow Data Preparation Checklist is a healthcare form used by researchers to ensure accurate reporting of participant progression through study stages.
pdfFiller scores top ratings on review platforms




Who needs Flow Data Checklist?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Flow Data Checklist
What is the Participant Flow Data Preparation Checklist?
The Participant Flow Data Preparation Checklist is a critical tool in medical research, designed to ensure accurate participant tracking and reporting throughout various stages of a study. This checklist aids in maintaining meticulous documentation of participant flow, encompassing all necessary elements to capture the comprehensive journey of participants from pre-assignment to study completion.
This checklist includes vital components such as study design outlines, arms or groups involved, and reasons for non-completion. By addressing these specifics, the checklist serves as a foundation for effective participant management, ultimately enhancing the integrity of the research outcomes.
Purpose and Benefits of the Participant Flow Data Preparation Checklist
The primary purpose of the Participant Flow Data Preparation Checklist is to support researchers in maintaining accurate records, which are paramount for regulatory compliance and achieving effective study outcomes. The checklist streamlines data preparation processes, thereby improving reporting accuracy and facilitating adherence to NIH participant flow guidelines.
Additionally, researchers gain significant benefits from this checklist, including enhanced organization and clarity during data collection. By utilizing the checklist, research teams can navigate complex documentation requirements with greater efficiency.
Who Needs the Participant Flow Data Preparation Checklist?
The checklist is essential for a variety of stakeholders in the medical research field. Key personnel such as clinical researchers, regulatory compliance officers, and study coordinators are the primary users who benefit from its structured approach.
-
Clinical researchers utilize the checklist to ensure adherence to study protocols.
-
Regulatory compliance officers require it to fulfill specific industry standards.
-
Study coordinators leverage the checklist for streamlined participant management.
In effect, using the checklist supports various roles within medical research, promoting a unified approach to participant flow documentation.
Key Features of the Participant Flow Data Preparation Checklist
The Participant Flow Data Preparation Checklist encompasses several critical components crucial for detailed participant tracking. It provides a conceptual overview of the study design and includes participant group descriptions to ensure all necessary data is captured effectively.
-
Detailed sections for conceptual overviews and participant group categorizations.
-
User-friendly design with interactive checkboxes and fields for data entry.
-
Each section is crafted to ensure comprehensive and systematic data collection.
These features collectively enhance the usability and effectiveness of the checklist, making it a vital resource for medical research participants.
How to Fill Out the Participant Flow Data Preparation Checklist Online (Step-by-Step)
To efficiently complete the checklist using pdfFiller, follow these step-by-step instructions:
-
Access pdfFiller and select the Participant Flow Data Preparation Checklist template.
-
Fill in specific fields, including study design and participant group information.
-
Ensure all required sections are completed for comprehensive documentation.
Additionally, double-check the information entered to guarantee accuracy and completeness before final submission. This attention to detail will enhance the quality of the data provided.
Common Errors and How to Avoid Them When Filling Out the Checklist
While completing the checklist, users may encounter common pitfalls that can result in incomplete submissions or misinterpretation of requirements. To mitigate these errors, consider the following recommendations:
-
Double-check each entry to ensure no fields are left incomplete.
-
Read through descriptions carefully to understand what is required in each section.
Implementing these strategies will significantly reduce the likelihood of mistakes, ultimately leading to more accurate and compliant documentation.
Security and Compliance for the Participant Flow Data Preparation Checklist
Handling sensitive research data securely is paramount. pdfFiller ensures the confidentiality and integrity of information through state-of-the-art encryption and adherence to stringent compliance standards, such as HIPAA and GDPR.
This commitment to security reaffirms the platform's dedication to protecting user data, allowing researchers to focus on their studies with confidence that their information remains safe throughout the documentation process.
How to Submit the Participant Flow Data Preparation Checklist
Once the checklist is completed, users have several submission options available to them. These methods may include:
-
Online submission directly through pdfFiller.
-
Email submission to relevant stakeholders.
-
Physical submission if necessary, according to institutional requirements.
Be mindful of any deadlines or processing times associated with the selected submission method to ensure timely processing of the checklist.
What Happens After You Submit the Participant Flow Data Preparation Checklist?
After submission, the checklist undergoes a review and approval process. It is essential to understand the expected outcomes and how to track the status of the submission. Users should prepare for potential follow-up actions, such as clarifications or amendments required by the reviewing body.
Get Started with pdfFiller for Your Participant Flow Data Preparation Checklist
Utilizing pdfFiller can greatly enhance your experience while completing the Participant Flow Data Preparation Checklist. The platform simplifies the filling process with its intuitive interface, enabling you to focus on gathering essential participant data effortlessly.
Explore the extensive range of editing and form-filling capabilities offered by pdfFiller to optimize your medical documentation efforts.
How to fill out the Flow Data Checklist
-
1.To access the Participant Flow Data Preparation Checklist on pdfFiller, visit the pdfFiller website and use the search bar to find the form by its name. Click on the link to open the form.
-
2.Once the form is open, use the pdfFiller interface to navigate through the document. Click on each field to start filling out your information.
-
3.Before you begin, gather all necessary information, such as study design details, participant group specifics, and any data regarding reasons for non-completion.
-
4.As you fill out the form, ensure that you check each relevant box and enter required information in the fields provided. Review the 'Explanation' section for guidance on specific elements to include.
-
5.Once you have completed all sections, take a moment to review the entire form for any missing information or errors. Make any necessary corrections before finalizing.
-
6.After reviewing, you can save your work directly on pdfFiller or choose to download the completed form. Consider exporting it as a PDF for easier sharing.
-
7.Finally, submit the form as required by your study protocols or institutional guidelines. Use the submission options on pdfFiller to send the document electronically or print it out for manual submission.

Who should fill out the Participant Flow Data Preparation Checklist?
This checklist should be filled out by clinical researchers, study coordinators, and regulatory affairs personnel involved in medical research projects to ensure accurate documentation of participant flow.
Is there a deadline for completing the checklist?
Deadlines for completing the checklist will depend on your specific study timeline. It is vital to complete it before submitting participant data for review to ensure compliance with research protocols.
What submission methods are accepted for this checklist?
The checklist can typically be submitted electronically through your research institution's designated platform. Alternately, you can print it and submit it as a hard copy if required.
Are there any supporting documents required with the checklist?
Usually, there are no supporting documents required specifically for the checklist itself. However, ensure that you have associated study protocol documents ready in case they are requested.
What are common mistakes to avoid while filling the checklist?
Common mistakes include leaving mandatory fields blank, failing to check appropriate boxes, and not providing complete participant details. Carefully review to avoid these issues.
How long does it take to process the checklist after submission?
Processing times can vary based on the institution's policies. Generally, you can expect feedback within a few weeks, but confirm with your research office for specific timelines.
What specific details are essential to include in this checklist?
Essential details include the conceptual overview of the study design, the number of groups or arms, periods of research, and reasons for non-completion to support thorough data analysis.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.






















