Last updated on Nov 6, 2015

Get the free Investigational New Drug Application (IND)
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is IND Application
The Investigational New Drug Application (IND) is a healthcare form used by drug sponsors to request FDA permission for clinical trials of new drugs.
pdfFiller scores top ratings on review platforms




Who needs IND Application?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to IND Application
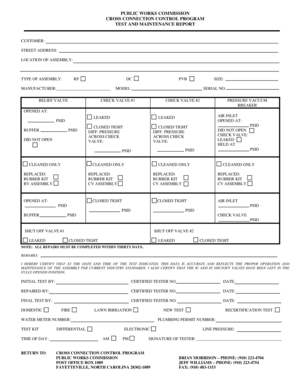
What is the Investigational New Drug Application (IND)?
The Investigational New Drug Application (IND) serves as a crucial form in the drug development process, requesting permission from the FDA to conduct clinical trials for new drugs. This application outlines fundamental information, such as the sponsor's details, the clinical investigation plan, and the drug's intended use.
Key components of the IND application include:
-
Complete sponsor information, including contact details.
-
A detailed clinical investigation plan explaining the study's methodology.
-
A description of the drug, including its chemical composition and intended therapeutic effects.
Obtaining FDA approval through the IND is essential for the initiation of clinical trials, ensuring compliance with regulatory standards designed to protect participant safety and validate drug efficacy.
Purpose and Benefits of the Investigational New Drug Application (IND)
The IND application is a critical document that facilitates the initiation of clinical trials for new drugs. By ensuring regulatory oversight, it offers significant advantages to drug sponsors, enhancing the safety and effectiveness of clinical research.
Benefits of filing an IND include:
-
Streamlined initiation of clinical trials through structured application processes.
-
Regulatory oversight that promotes patient safety during experiments.
-
Enhanced transparency in drug development, fostering public trust.
These advantages underscore the necessity of the IND for ensuring a thorough evaluation of the investigational drug.
Who Needs to File the Investigational New Drug Application (IND)?
The IND application must be filed by sponsors and their authorized representatives who are responsible for developing new drugs. Typically, these are organizations such as pharmaceutical companies and biotechnology firms focusing on innovative treatment options.
Key roles involved in the IND process include:
-
Sponsors who assume legal responsibility for the drug.
-
Authorized representatives who submit the IND on behalf of the sponsor.
Ensuring that the IND is submitted by eligible organizations is vital for compliance with FDA regulations.
How to Fill Out the Investigational New Drug Application (IND) Online
Completing the IND application online through pdfFiller is an efficient process that streamlines form filling and ensures compliance with required standards. The platform provides several features that assist users during this process.
To fill out the IND application, follow these steps:
-
Access the IND application form on pdfFiller.
-
Enter required information in the fields, including sponsor name and submission date.
-
Review each section carefully to ensure all blanks are addressed.
Utilizing pdfFiller’s practical tools enhances the likelihood of successfully completing the necessary entries.
Common Errors and How to Avoid Them in the Investigational New Drug Application (IND)
Filing the IND application can be complex, and various common errors often impede successful submission. Awareness of these mistakes can save time and ensure compliance with the FDA guidelines.
Common error-prone sections include:
-
Incorrect sponsor information entry.
-
Missing required signatures in submission sections.
Implementing best practices for reviewing the form before submission is crucial. Leveraging validation tools in pdfFiller can also significantly reduce the risk of overlooking critical information.
How to Sign and Submit the Investigational New Drug Application (IND)
Understanding the signing and submission process for the IND application is essential for compliance with FDA requirements. The submission can be done through various methods, depending on the type of signature used.
To properly sign and submit the IND, follow these guidelines:
-
Utilize a digital signature if applicable to your submission type.
-
Ensure the form is completed entirely before submission.
-
Track your submission through the FDA’s acknowledgement processes.
These practices enhance the reliability of your submission and help in managing communication with the FDA.
What Happens After You Submit the Investigational New Drug Application (IND)?
After submitting the IND application, it enters a review process conducted by the FDA. The timeline for receiving a response can vary depending on several factors, including the complexity of the application.
Key points to consider post-submission include:
-
Anticipating the review timelines and planning accordingly.
-
Regularly checking your application status with the FDA.
-
Being prepared to respond promptly to requests for additional information or amendments.
Understanding these steps can help manage expectations during the approval process.
Security and Compliance for the Investigational New Drug Application (IND)
Document security and compliance with regulations are critical when handling the IND application. Protecting sensitive information throughout the submission process is paramount.
Key security measures include:
-
Utilizing data protection measures relevant to IND submissions.
-
Ensuring compliance with HIPAA and GDPR standards for privacy.
pdfFiller employs robust security practices, including encryption, to safeguard confidential documents during the IND submission process.
Leverage pdfFiller for Your Investigational New Drug Application (IND)
For an efficient and user-friendly IND application process, pdfFiller offers exceptional features tailored to meet the needs of users. Utilizing this cloud-based platform can enhance the overall experience when completing healthcare forms.
Advantages of using pdfFiller include:
-
Ease of filling out forms and eSigning.
-
Cloud-based document management that simplifies organization.
Testimonials from satisfied users further demonstrate the efficacy of pdfFiller in successfully completing IND applications.
How to fill out the IND Application
-
1.Access pdfFiller and search for the 'Investigational New Drug Application (IND)' form in the available templates.
-
2.Open the form to view the complete layout that includes all necessary sections for completion.
-
3.Before starting, gather essential information such as the name of the sponsor, drug details, and clinical investigation plan.
-
4.Begin filling in the required fields, including the 'Name of Sponsor', 'Date of Submission', and checkboxes for type of submission.
-
5.Utilize pdfFiller's interface to navigate through the form efficiently by clicking on fields to enter information directly.
-
6.Double-check each section for accuracy and completeness as you progress through the form.
-
7.Once all fields are completed, review the entire form in pdfFiller to ensure all information is correctly entered and properly formatted.
-
8.Finalize the form by clicking on the 'Save' option to keep a copy in your pdfFiller account.
-
9.If required, download the filled form in your preferred format or use the provided options to submit it electronically directly through pdfFiller.

Who is eligible to submit the IND application?
Eligibility to submit the IND application typically includes pharmaceutical companies, research organizations, and clinical trial sponsors that intend to conduct clinical trials for a new investigational drug.
What is the typical processing time for an IND application?
The processing time for an IND application can vary, but it generally takes 30 days for the FDA to review the application for safety concerns before approving clinical trial initiation.
Are there any specific documents required when submitting the IND?
Yes, required documents usually include the investigational plan, drug information, and any informed consent documents. Ensure all supporting documents are prepared before submission.
What common mistakes should be avoided when completing the IND form?
Common mistakes include failing to complete all required fields, inaccurate information regarding the drug or sponsor, and not including necessary supporting documents. Double-checking details can prevent delays.
How do I submit the IND application once completed?
Once the IND application is completed, it can be submitted via the FDA's electronic submission system, or if required, you can use pdfFiller to download and submit the form as directed by the FDA.
Are there any fees associated with submitting the IND application?
Fees for submitting an IND application vary; however, typically there are no application fees. It is recommended to verify with the FDA for any changes in policy regarding fees.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.