Last updated on Nov 10, 2015

Get the free Biosafety Questionnaire for Flow Cytometry Facilities
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Biosafety Questionnaire
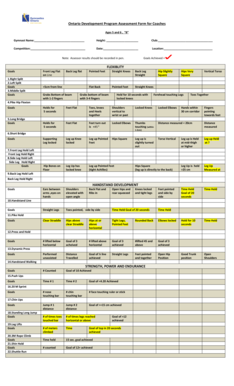
The Biosafety Questionnaire for Flow Cytometry Facilities is a healthcare form used by laboratory directors to gather essential information regarding samples in flow cytometry core facilities.
pdfFiller scores top ratings on review platforms




Who needs Biosafety Questionnaire?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Biosafety Questionnaire
What is the Biosafety Questionnaire for Flow Cytometry Facilities?
The Biosafety Questionnaire for Flow Cytometry Facilities is a critical tool in ensuring safety and compliance within flow cytometry core facilities. This questionnaire gathers essential information regarding samples, potential risks, and the safety measures in place, helping to mitigate any hazards that may arise during experiments. By completing this healthcare form, facilities can better manage the risks associated with various biological materials.
Purpose and Benefits of the Biosafety Questionnaire for Flow Cytometry Facilities
This laboratory safety form serves multiple purposes, primarily by aiding in the maintenance of laboratory safety and regulatory compliance. Obtaining necessary biosafety approvals prior to initiating experiments is a vital requirement, ensuring that all safety protocols are in place. The completion of this research safety form allows labs to identify potential hazards and implement appropriate safety measures to protect both personnel and the research environment.
Key Features of the Biosafety Questionnaire for Flow Cytometry Facilities
The Biosafety Questionnaire contains several key components necessary for comprehensive biosafety evaluations. Key fields in the questionnaire include:
-
Sample sources and types
-
Identification of infectious agents
-
Biosafety level categorizations
-
Inactivation methods
-
Blood-borne pathogen screening
Importantly, a signature from the laboratory director is required for validation, along with other essential confirmations to ensure the integrity of the information provided.
Who Needs the Biosafety Questionnaire for Flow Cytometry Facilities?
Essential stakeholders in this process include laboratory directors and principal investigators who must ensure the safekeeping and ethical handling of samples. Various research projects specifically necessitate the completion of this form, especially those involving human samples or infectious agents. By engaging those responsible for overseeing laboratory activities, compliance with biosafety protocols is enhanced.
How to Fill Out the Biosafety Questionnaire for Flow Cytometry Facilities Online (Step-by-Step)
Filling out the biosafety questionnaire online is straightforward using pdfFiller. Follow these steps for efficient completion:
-
Access the questionnaire through the pdfFiller platform.
-
Fill in required fields, such as sample information and risk assessments.
-
Add details regarding biosafety levels and inactivation methods.
-
Review the information for completeness and accuracy.
-
Obtain the laboratory director's signature before submission.
This approach ensures an accurate and efficient workflow, reducing potential errors in submission.
Common Errors and How to Avoid Them
When completing the biosafety questionnaire, various common errors may arise. These include omissions of required information or misclassification of sample types. To minimize inaccuracies, consider the following tips:
-
Review each field for completeness before finalizing.
-
Double-check entries against standard biosafety protocols.
-
Consult with colleagues if uncertainties about classifications exist.
Taking these precautions will contribute greatly to the reliability of the submitted form.
Digital Signature Requirements for the Biosafety Questionnaire
The laboratory director’s signature is a vital component of the biosafety questionnaire, affirming that all safety measures are understood and implemented. pdfFiller accepts digital signatures, which eases the signing process while maintaining document integrity. The platform incorporates robust security measures to protect sensitive information during the signing process, ensuring compliance with necessary regulations.
Submission Methods and Follow-Up After Completing the Biosafety Questionnaire
Once the biosafety questionnaire is completed, it can be submitted through various channels, such as online submission directly via pdfFiller or by printing and mailing it. After submission, tracking the status of your questionnaire is essential, as it provides clarity on approvals and any additional requirements that might arise.
Security and Compliance Considerations for the Biosafety Questionnaire
Data protection is crucial when dealing with biosafety information. pdfFiller prioritizes privacy and compliance with security standards such as HIPAA and GDPR, employing advanced encryption to safeguard all data. Adhering to these security measures ensures that sensitive information is processed safely, maintaining the confidentiality of research materials.
Empower Your Flow Cytometry Research with pdfFiller's Online Form Capabilities
Utilizing pdfFiller's platform simplifies the completion and management of the biosafety questionnaire, streamlining the biosafety approval process effectively. With its user-friendly interface and robust document management capabilities, researchers can achieve compliance while enhancing the overall efficiency of flows in their laboratory operations.
How to fill out the Biosafety Questionnaire
-
1.To access the Biosafety Questionnaire for Flow Cytometry Facilities, visit pdfFiller and log into your account.
-
2.Use the search function to find the form by entering its name: 'Biosafety Questionnaire for Flow Cytometry Facilities'.
-
3.Once located, click on the form to open it in the pdfFiller interface.
-
4.Review the form’s structure to familiarize yourself with its sections, including sample sources and biosafety details.
-
5.Gather all necessary information such as sample sources, any potential infectious agents, biosafety levels, and inactivation methods before starting to fill out the form.
-
6.Begin completing the fillable fields, ensuring that you provide accurate information in each section.
-
7.Utilize checkboxes where applicable to indicate your responses clearly.
-
8.Refer to the description and explanation sections within the form for guidance on specific entries.
-
9.As you complete the form, save your progress regularly to avoid losing any data.
-
10.Once all fields are complete, review the form carefully to ensure all information is accurate and that you have added your signature.
-
11.Finalize the form by clicking on the 'Save' option, then select your preferred method to download or submit the form directly through pdfFiller.

Who is required to sign the Biosafety Questionnaire?
The Biosafety Questionnaire must be signed by the laboratory director or principal investigator before any projects or experiments commence.
What information is needed to complete the form?
You will need to gather details on sample sources, potential infectious agents, biosafety levels, blood-borne pathogen screening, and any fixation protocols before filling out the form.
Are there any notarization requirements for this form?
No, the Biosafety Questionnaire does not require notarization; it only needs to be signed by the laboratory director.
How do I submit the completed questionnaire?
After completing and reviewing the form on pdfFiller, you can download it or submit it directly through the platform, following the provided submission instructions.
What common mistakes should I avoid while filling out the form?
Ensure to double-check all provided information, avoid leaving any required fields blank, and remember to sign the document before submission.
Is there a deadline for submitting this form?
While specific deadlines may not be stated, it is crucial to complete and submit the form before initiating any related projects or experiments to ensure compliance.
How can I access additional support if needed?
For any assistance while completing the form, you can refer to pdfFiller's help center or reach out to your institution's biosafety officer for guidance.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.