Last updated on Dec 7, 2015

Get the free Agendia FFPE Block Submission Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Agendia FFPE Form
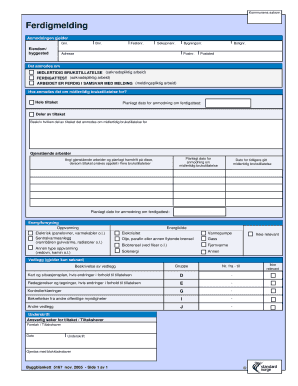
The Agendia FFPE Block Submission Form is a medical document used by healthcare professionals to submit FFPE tumor blocks and unstained slides for molecular testing.
pdfFiller scores top ratings on review platforms




Who needs Agendia FFPE Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Agendia FFPE Form
What is the Agendia FFPE Block Submission Form?
The Agendia FFPE Block Submission Form is a crucial document used for submitting formalin-fixed paraffin-embedded (FFPE) tumor blocks and unstained slides essential for Agendia's molecular testing. This form facilitates the accurate collection of specimens and ensures that they are processed correctly, leading to reliable test results. As a vital component of the testing protocol, the Agendia FFPE Block Submission Form plays a significant role in supporting patient diagnoses and treatment decisions.
Purpose and Benefits of the Agendia FFPE Block Submission Form
The primary purpose of the Agendia FFPE Block Submission Form is to standardize the submission process for medical specimens. Utilizing this form provides numerous benefits, including enhanced accuracy in test results due to consistent specimen handling protocols. Furthermore, the standardized format simplifies the submission process, allowing healthcare providers to efficiently send specimens for analysis.
Key Features of the Agendia FFPE Block Submission Form
The form includes several essential features designed to streamline the submission process. Key components consist of:
-
Fillable fields for easy data entry.
-
Instructions on how to label, package, and ship specimens adequately.
-
Specific requirements for specimen barcode labels and unstained slide preparations.
These features work together to ensure that all necessary information is accurately captured, minimizing errors during submission.
Who Needs the Agendia FFPE Block Submission Form?
The primary users of the Agendia FFPE Block Submission Form include pathologists responsible for specimen submission and patients providing authorization for testing. Scenarios that necessitate its use typically involve scenarios where molecular testing is indicated for tumor assessment. Patient authorization is crucial in this process, ensuring compliance with health regulations and personal privacy.
How to Fill Out the Agendia FFPE Block Submission Form Online (Step-by-Step)
Filling out the Agendia FFPE Block Submission Form online is straightforward when following these steps:
-
Access the form through the designated platform.
-
Carefully enter the required information in each field.
-
Ensure to apply one specimen barcode label as specified.
-
Double-check all entries for clarity and correctness before submission.
-
Submit the form digitally or prepare it for physical shipping based on your needs.
By following these steps, users can avoid common errors that may arise during the filling process.
Required Documents and Supporting Materials
To ensure a complete submission, the following documents must accompany the Agendia FFPE Block Submission Form:
-
The completed Agendia test request form.
-
Any relevant medical records necessary for patient consent.
-
A checklist of items to confirm all documentation is included.
Having these documents prepared beforehand will facilitate a smoother submission process.
Submission Methods and Delivery for the Agendia FFPE Block Submission Form
There are several methods available for submitting the completed form and specimens:
-
Online submission through a secured portal.
-
Mailing the physical copies to Agendia's laboratory.
-
Contacting Agendia for specific delivery options based on location.
After submission, users can expect a notification regarding the status of their specimens and the testing process.
Security and Compliance for Submitting Medical Forms
Data security is paramount when handling health records. pdfFiller employs multiple security measures such as 256-bit encryption and is fully compliant with HIPAA regulations to protect users' sensitive information. Furthermore, privacy considerations are integrated throughout the form-filling and submission processes, ensuring personal data remains confidential and secure.
How pdfFiller Can Assist You with the Agendia FFPE Block Submission Form
pdfFiller offers a variety of capabilities to assist users in filling out the Agendia FFPE Block Submission Form efficiently. Users can benefit from features like:
-
Text and image editing for customization.
-
eSigning options for secure document approval.
-
Cloud storage for easy access and management of forms.
This platform streamlines the entire process, making it easier for users to complete and submit forms accurately.
Next Steps After Submitting the Agendia FFPE Block Submission Form
After submitting the Agendia FFPE Block Submission Form, users can expect to receive confirmation within a specified timeframe. It's important to check the status of the application regularly and understand the protocols for handling any potential rejections. In case re-submission is necessary, users should follow the indicated renewal process for efficient resolution.
How to fill out the Agendia FFPE Form
-
1.To begin, access the Agendia FFPE Block Submission Form on pdfFiller by searching for the form name in the platform's search bar.
-
2.Once the form is open, navigate through the document's sections using pdfFiller's intuitive interface.
-
3.Before filling out the form, gather necessary information including patient details, tumor specimen specifics, and your completed Agendia test request form.
-
4.Start filling out the required fields, ensuring all information is accurate and clearly presented.
-
5.Use the 'Apply one specimen barcode label' field to appropriately label each specimen as per Agendia's instructions.
-
6.Fill in the section for preparing ten 5μm serial unstained slides, ensuring that all details match the specifications outlined in the instructions.
-
7.Check that you have included a completed or printed Agendia test request form with your submission materials.
-
8.Once you’ve filled out all relevant sections of the form, review the information for completeness and correctness.
-
9.After finalizing the form, use pdfFiller's options to save your completed form, download it as a PDF, or submit directly through the platform.

Who is eligible to use the Agendia FFPE Block Submission Form?
Healthcare professionals, such as pathologists, and patients needing molecular testing are eligible to use the Agendia FFPE Block Submission Form. It is intended for submissions of FFPE tumor blocks and unstained slides.
Are there any deadlines for submitting the submission form?
While specific deadlines are not mentioned, it's essential to submit the Agendia FFPE Block Submission Form as soon as possible after specimen preparation to ensure timely testing and processing.
What methods are available for submitting the completed form?
The completed Agendia FFPE Block Submission Form can be submitted through pdfFiller by directly sending it from the platform or downloading it to send via traditional mail, depending on your needs.
What supporting documents are required with the submission?
In addition to the Agendia FFPE Block Submission Form, you must include a completed Agendia test request form and ensure that all tumor specimens are accurately labeled and packaged.
What are some common mistakes to avoid when filling out the form?
Common mistakes include missing required fields, providing incorrect specimen details, and not including the Agendia test request form. Double-check all information before submission to avoid delays.
How long does processing take after submission?
Processing times can vary based on the laboratory's workload, but it is advised to contact Agendia directly for the most accurate estimates regarding specific test turnaround times.
What specific information is needed for the form?
You will need patient information, details of the tumor specimens being submitted, and to ensure a completed Agendia test request form is prepared as part of your submission.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.