Last updated on Jan 20, 2016
Get the free ESMO Clinical Benefit Scale Evaluation Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is ESMO Evaluation Form
The ESMO Clinical Benefit Scale Evaluation Form is a medical document used by healthcare professionals to assess the clinical benefits of therapies primarily focused on progression-free survival (PFS).
pdfFiller scores top ratings on review platforms




Who needs ESMO Evaluation Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to ESMO Evaluation Form
What is the ESMO Clinical Benefit Scale Evaluation Form?
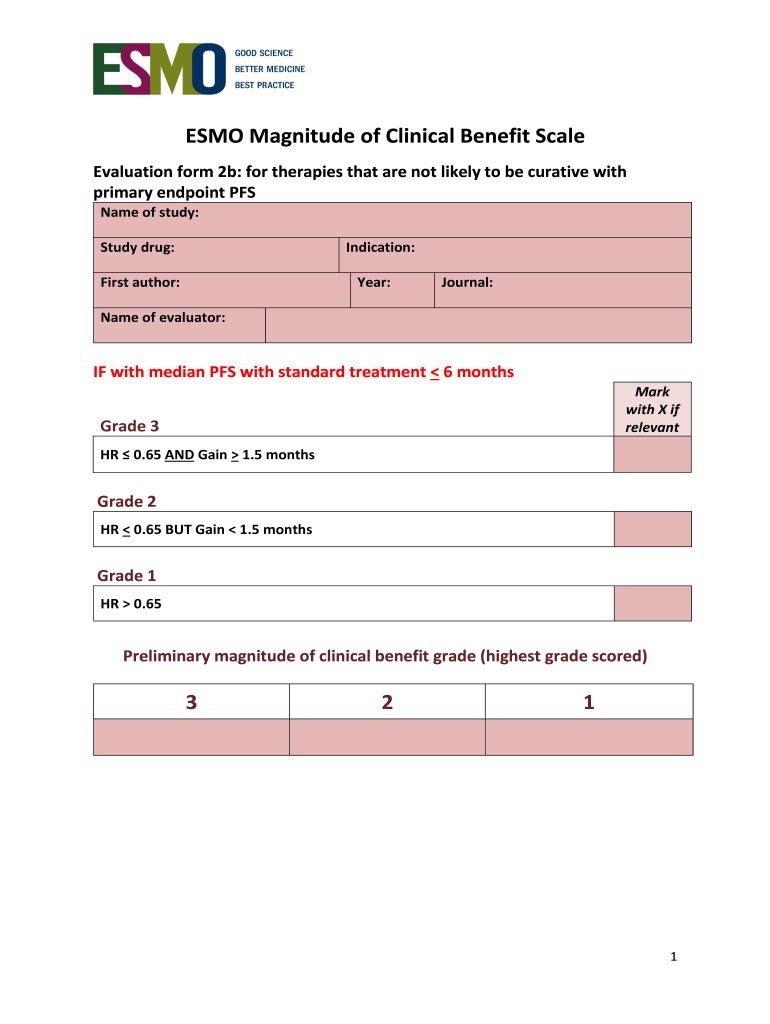
The ESMO Clinical Benefit Scale Evaluation Form is a critical tool in the evaluation of non-curative therapies in clinical settings. It serves to assess the magnitude of clinical benefit, focusing primarily on progression-free survival (PFS) as a primary endpoint. This evaluation form enables healthcare professionals to systematically review treatment outcomes related to various therapies.
By utilizing the ESMO evaluation form, practitioners can make informed decisions regarding therapy efficacy and patient management. The precise definition of this form reflects its role as a structured method of quantifying clinical benefit, ensuring consistency and reliability in evaluations.
Purpose and Benefits of the ESMO Clinical Benefit Scale Evaluation Form
The purpose of the ESMO Clinical Benefit Scale Evaluation Form is to provide healthcare professionals with a clear framework for measuring clinical benefit during therapy evaluation. This structured scoring system enhances the clarity and accuracy of patient assessments, streamlining communication among clinical teams.
Its benefits include improved patient care and better-informed treatment decisions, as it allows for the quantification of clinical outcomes. The form aids in ensuring that the care provided is aligned with patients' needs, specifically regarding their journey through therapies with non-curative intent.
Key Features of the ESMO Clinical Benefit Scale Evaluation Form
The ESMO Clinical Benefit Scale Evaluation Form contains several unique features designed to facilitate effective evaluation and documentation. Firstly, it includes fillable patterns like checkboxes and labeled input fields, which enhance user-friendliness.
-
Sections dedicated to study details, toxicity assessments, and quality of life evaluations help ensure comprehensive documentation.
-
The scoring system integration assesses hazards ratios and PFS gain, allowing for a thorough analysis of treatment efficacy.
-
These features make it easier for healthcare providers to implement a standardized approach to evaluating non-curative therapies.
Who Needs the ESMO Clinical Benefit Scale Evaluation Form?
The primary users of the ESMO Clinical Benefit Scale Evaluation Form include healthcare professionals and clinical trial researchers. Its integration into clinical settings is essential for accurate therapy assessment.
This form is particularly beneficial when assessing patients in scenarios requiring detailed clinical trial assessments or structured medical evaluations. It allows users to systematically track therapy effectiveness and make informed decisions based on clinical data.
How to Fill Out the ESMO Clinical Benefit Scale Evaluation Form Online
To complete the ESMO Clinical Benefit Scale Evaluation Form using pdfFiller, follow these step-by-step instructions:
-
Access the form online through pdfFiller’s platform.
-
Fill in study details accurately, including title and involved parties.
-
Complete sections on toxicity and quality of life using the provided fields.
-
Ensure all information is accurate and complete before submission.
-
Utilize pdfFiller’s tools for checking and maintaining the integrity of your entries.
Common Errors and How to Avoid Them When Using the ESMO Clinical Benefit Scale Evaluation Form
Users may encounter several common errors when filling out the ESMO Clinical Benefit Scale Evaluation Form. These errors can include incomplete fields and incorrect scoring, which may lead to inaccurate evaluations.
-
Ensure all necessary fields are filled to avoid submission delays.
-
Double-check scoring to maintain consistency with established guidelines.
-
Validation of entries is crucial for ensuring the accuracy of assessments.
Submitting the ESMO Clinical Benefit Scale Evaluation Form: Methods and Best Practices
Proper submission of the completed ESMO Clinical Benefit Scale Evaluation Form is essential for compliance and acceptance. Users can submit the form through various methods:
-
Online submission via pdfFiller is recommended for efficiency.
-
For physical submissions, ensure the form is printed clearly and completely filled.
-
Confirm receipt of the submitted form, and track the status for peace of mind.
Security and Compliance when Using the ESMO Clinical Benefit Scale Evaluation Form
When handling the ESMO Clinical Benefit Scale Evaluation Form, security measures and compliance standards are paramount. pdfFiller employs 256-bit encryption to safeguard user data and maintain privacy.
Compliance with regulations such as HIPAA and GDPR ensures that sensitive information is processed securely. Users can trust that their data is well protected throughout the evaluation process.
How pdfFiller Supports You in Completing the ESMO Clinical Benefit Scale Evaluation Form
pdfFiller significantly enhances the user experience when completing the ESMO Clinical Benefit Scale Evaluation Form. Its capabilities include editing, eSigning, and sharing documents easily, making the entire process efficient.
By using pdfFiller, users benefit from convenient tools for creating and filling out forms, ensuring a streamlined experience for healthcare providers managing evaluations.
How to fill out the ESMO Evaluation Form
-
1.To access the ESMO Clinical Benefit Scale Evaluation Form, visit pdfFiller's website and use the search function to locate the form by its official name.
-
2.After opening the form, familiarize yourself with the interface. Note the fillable fields such as checkboxes and input boxes for entering study details.
-
3.Before filling out the form, gather all necessary information including the patient's medical history, treatment details, and data related to toxicity and quality of life assessments.
-
4.Start by filling in the study details section, ensuring you provide all requested information accurately. Use checkboxes where applicable to simplify responses.
-
5.Proceed to the toxicity assessment section, carefully assessing and selecting the appropriate responses based on the therapy's impact on the patient.
-
6.Next, complete the quality of life evaluation section by referencing any quality of life metrics you’ve collected throughout the treatment.
-
7.Once you've completed all sections of the form, review it thoroughly for any errors or omissions. Make sure all fields are filled correctly.
-
8.Finalize the form by saving your changes. You can download the completed form in various formats or submit it directly through pdfFiller if required.

Who is eligible to use the ESMO Clinical Benefit Scale Evaluation Form?
The form is primarily designed for healthcare professionals, including oncologists and researchers, who are evaluating the effectiveness of non-curative therapies on patient outcomes.
What is the deadline for submitting the evaluation form?
Deadlines for submission may vary depending on clinical trial protocols or evaluation timelines set by institutions, so it's important to check with relevant authorities for specific dates.
How should I submit the completed ESMO form?
You can submit the completed form via email, physical mail, or directly through designated platforms, depending on the requirements of the trial or study overseeing authority.
Are there any specific documents required when filling out the ESMO Evaluation Form?
Typically, you will need supporting documents related to the patient’s treatment history and any prior evaluations to provide a comprehensive assessment and ensure accuracy.
What are common mistakes to avoid when filling the form?
Ensure that all required fields are filled out and double-check for any incorrect entries. Be careful with numerical values and ensure you follow the scoring guidelines correctly.
How long does it take to process the ESMO Clinical Benefit Scale Evaluation Form?
Processing times can vary based on the organization, but typically expect a few weeks for results to be assessed and communicated following submission.
What sections are included in the ESMO Evaluation Form?
The form includes sections for study details, toxicity assessments, and quality of life evaluations, each aimed at determining the clinical benefit of the treatments in question.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.