Last updated on Jan 26, 2016

Get the free INTERGROWTH-21st Fetal Growth Adverse Event Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Fetal Growth AE Form
The INTERGROWTH-21st Fetal Growth Adverse Event Form is a medical document used by healthcare professionals to document and track adverse events during fetal growth studies.
pdfFiller scores top ratings on review platforms




Who needs Fetal Growth AE Form?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Fetal Growth AE Form
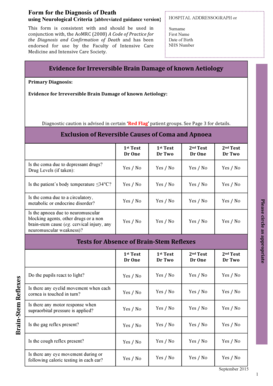
What is the INTERGROWTH-21st Fetal Growth Adverse Event Form?
The INTERGROWTH-21st Fetal Growth Adverse Event Form is a pivotal document used in fetal growth studies to record and track adverse events associated with pregnancies. This form plays a crucial role in maintaining the integrity of research by providing a standardized method for healthcare professionals to document issues that may arise during the study. Proper documentation of these adverse events is essential for ensuring patient safety and advancing clinical research.
Purpose and Benefits of the INTERGROWTH-21st Fetal Growth Adverse Event Form
This fetal growth study form is fundamental for healthcare providers as it enhances research quality by consistently capturing vital information related to adverse events. Its use contributes significantly to patient safety by enabling timely responses to any complications that may occur. Healthcare professionals benefit from this form as it standardizes the collection of data, which is critical for assessing the effects of different interventions and ensuring compliance with research protocols.
Key Features of the INTERGROWTH-21st Fetal Growth Adverse Event Form
The INTERGROWTH-21st Fetal Growth Adverse Event Form is structured to facilitate accurate documentation through various required fields. The form includes:
-
Checkboxes for quickly identifying types of adverse events.
-
Blank fields to provide detailed descriptions of events and actions taken.
-
Sections for demographic data, including study subject number and clinic code.
This design ensures that all pertinent information is captured succinctly and systematically.
Who Needs the INTERGROWTH-21st Fetal Growth Adverse Event Form?
This fetal growth study form is essential for a wide array of users, primarily healthcare professionals, including obstetricians, midwives, and researchers who are involved in fetal growth studies. Eligibility to use the form typically includes those participating in clinical trials or studies mandated to document adverse events. Scenarios that necessitate the form may include unexpected maternal or fetal complications that arise during the monitoring of pregnancies.
How to Fill Out the INTERGROWTH-21st Fetal Growth Adverse Event Form Online (Step-by-Step)
Completing the INTERGROWTH-21st Fetal Growth Adverse Event Form online is straightforward. Here’s how:
-
Access the form through the designated platform.
-
Enter the study subject number and relevant demographic information.
-
Select the type of adverse event using checkboxes.
-
Provide detailed descriptions in the blank fields provided.
-
Review the completed form for accuracy before submission.
This structured approach ensures all necessary information is captured effectively.
Digital Signature and Submission Process for the INTERGROWTH-21st Fetal Growth Adverse Event Form
When submitting the INTERGROWTH-21st Fetal Growth Adverse Event Form, signatures are required. Healthcare professionals can opt for either digital signatures or traditional wet signatures, depending on their preference or institutional requirements. Submissions can be made online for immediate processing or via traditional methods such as fax or mail to ensure compliance and security.
Security and Compliance for the INTERGROWTH-21st Fetal Growth Adverse Event Form
pdfFiller prioritizes security when handling sensitive patient information through robust measures like 256-bit encryption. Compliance with HIPAA and GDPR regulations is strictly adhered to, ensuring that confidentiality is maintained throughout the form's use. This focus on security is essential for the protection of medical data related to adverse events in fetal growth studies.
What Happens After You Submit the INTERGROWTH-21st Fetal Growth Adverse Event Form?
After submitting the INTERGROWTH-21st Fetal Growth Adverse Event Form, users can expect a confirmation of receipt and tracking information. The processing times for the submitted forms can vary depending on the institution, but typically, users will be informed about expected timelines and any further actions required after submission. This transparency is crucial for maintaining efficient communication between researchers and healthcare providers.
How to Download, Save, and Print the INTERGROWTH-21st Fetal Growth Adverse Event Form
Downloading, saving, and printing the INTERGROWTH-21st Fetal Growth Adverse Event Form can be accomplished in several simple steps:
-
Navigate to the digital form on the platform.
-
Select the download option, choosing your desired format.
-
Save the file to your device for future reference.
-
Print the form as needed for offline processing or archiving.
Various formats may be available for this form, allowing users to select the one that best suits their needs.
Explore pdfFiller for Your Document Management Needs
Utilizing pdfFiller enhances the process of filling out and editing forms. The platform offers user-friendly features that streamline the completion of medical records and consent forms while maintaining high levels of security. Whether it’s for eSignatures or editing capabilities, pdfFiller stands out as an efficient choice for managing important documents.
How to fill out the Fetal Growth AE Form
-
1.To access the INTERGROWTH-21st Fetal Growth Adverse Event Form on pdfFiller, navigate to the platform and use the search bar to locate the form by title.
-
2.Once you have found the form, click on it to open the document in the pdfFiller editing interface.
-
3.Before completing the form, gather all necessary information including the study subject number, date of birth, clinic code, antenatal record number, final diagnosis, timing of the event, actions taken, and outcomes.
-
4.Begin filling in the form by clicking on each blank field. Use the provided checkboxes to indicate relevant options, ensuring you provide accurate details.
-
5.Follow the clear instructions embedded in the form to correctly complete each section. If unsure, refer to guidance notes that may appear alongside the fields.
-
6.After entering all required information, review the form thoroughly to ensure all details are accurate and complete, minimizing the risk of errors.
-
7.Once satisfied with the form, use the save option to store your progress, download for your records, or submit directly through pdfFiller if necessary.

Who is eligible to fill out the INTERGROWTH-21st Fetal Growth Adverse Event Form?
Eligibility to fill out the form is primarily for healthcare professionals involved in fetal growth studies, particularly those documenting adverse events in clinical settings.
Is there a deadline for submitting the Fetal Growth Adverse Event Form?
While specific deadlines may depend on the study protocol, it is generally advisable to complete and submit the form as soon as possible after an adverse event occurs to ensure timely reporting.
How should I submit the INTERGROWTH-21st Fetal Growth Adverse Event Form?
The form can be submitted directly through pdfFiller if you use the platform. Alternatively, you can download it and submit it to the relevant study supervisor or research coordinator via email or physical submission.
What supporting documents are required when submitting this form?
Typically, no additional supporting documents are required with this form. However, you may need to include relevant medical records or notes related to the adverse event if deemed necessary.
What common mistakes should I avoid when filling out the form?
Common mistakes include omitting required information, providing inaccurate data, not following the instructions for completion, and failing to double-check for spelling and formatting errors.
How long does it take to process the submitted INTERGROWTH-21st Fetal Growth Adverse Event Form?
Processing times can vary depending on the research site's workflow, but typically, forms are reviewed within a few days to a week after submission.
What are the potential outcomes after submitting this form?
Outcomes after submission may include further investigation of the reported adverse event, adjustments to study protocols, or follow-up requests for additional information from the research team.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.