Create Clinical Trial Agreement with pdfFiller
Creating a Clinical Trial Agreement is crucial for ensuring compliance and clarity in clinical research protocols. With pdfFiller, users can effortlessly craft, edit, and manage agreements tailored to their specific needs.
What is a Clinical Trial Agreement?
A Clinical Trial Agreement (CTA) is a legally binding document that outlines the terms and conditions under which a clinical trial will be conducted. It details the responsibilities of the parties involved, the funding arrangements, data ownership, and confidentiality obligations, ensuring that all parties are aligned on the trial's scope and methodology.
How does a Clinical Trial Agreement improve document creation?
Having a standardized Clinical Trial Agreement streamlines document creation by providing clear guidelines and needed elements that must be included. This reduces errors and accelerates the approval process, thus facilitating smoother collaborations between sponsors and research institutions.
Finding and selecting existing templates for Clinical Trial Agreements
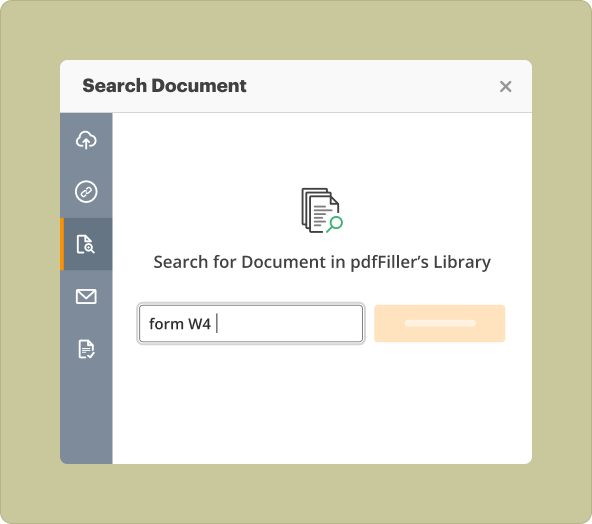
Users can locate numerous pre-existing templates for Clinical Trial Agreements within pdfFiller’s library. These templates are specifically designed to meet regulatory standards and presentations, allowing users to quickly choose and customize them according to their trial requirements.
Step-by-step: creating documents using Clinical Trial Agreement
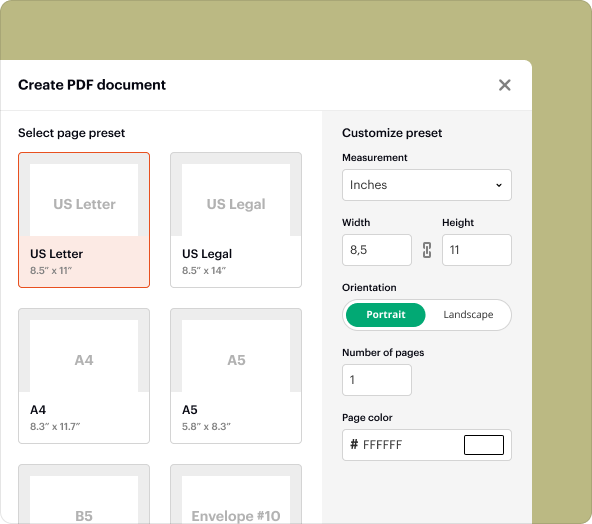
To create a Clinical Trial Agreement, follow these steps: 1. Log in to your pdfFiller account. 2. Navigate to the template library and search for 'Clinical Trial Agreement.' 3. Select a suitable template. 4. Customize the content fields with your trial details. 5. Save and prepare for sharing or collaboration.
-
1.Log in to pdfFiller
-
2.Access the template library
-
3.Search for Clinical Trial Agreement
-
4.Select and customize your template
-
5.Save the document
Customizing content and fields as you create
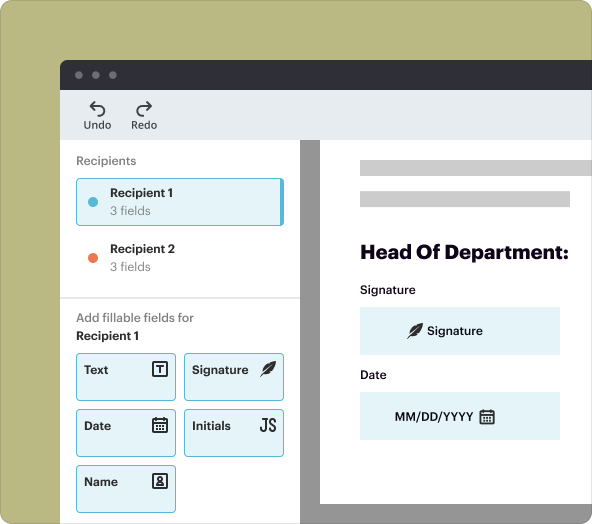
While customizing your Clinical Trial Agreement, you can modify text, add or remove fields, and adjust layouts. This flexibility ensures that all necessary clauses are included and that the document adheres to your trial's specifications.
-
1.Edit text for clarity and accuracy
-
2.Insert fields for signatures and dates
-
3.Adjust formatting as needed
Saving, organizing, and managing files after you create
pdfFiller allows users to save and organize their Clinical Trial Agreements in a structured manner. Categories or tags can be assigned for easier retrieval, ensuring that you can quickly access past documents related to various trials.
Sharing and collaborating on documents you create
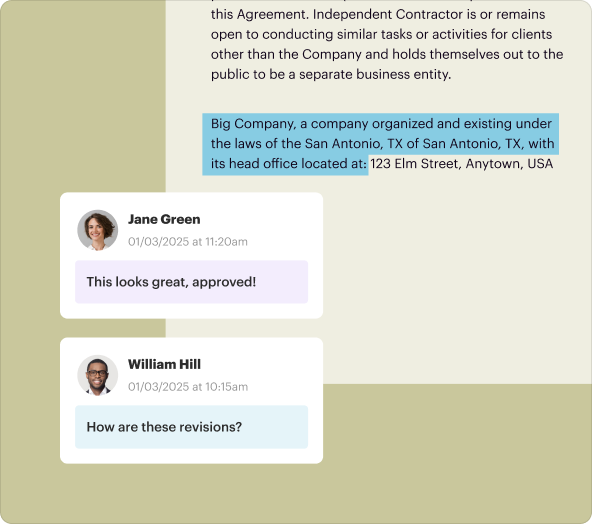
Once the Clinical Trial Agreement is finalized, it can easily be shared with all stakeholders for review and approval. Users can enable collaboration features to allow real-time editing and comments, enhancing the collective input on the document.
Typical cases and industries that often utilize Clinical Trial Agreements
Clinical Trial Agreements are predominantly used within the pharmaceutical, biotech, and medical device industries. They are pivotal in partnerships between academia and industry for clinical research and play a critical role in ensuring regulatory compliance.
-
1.Pharmaceutical research
-
2.Biotech partnerships
-
3.Medical device testing
-
4.Academic collaborations
These agreements are foundational in ensuring transparency and compliance across various stakeholders in clinical research.
Conclusion
In conclusion, using pdfFiller to create a Clinical Trial Agreement not only simplifies the document creation process but also provides essential tools for customization, management, and collaboration. Embracing this platform positions users to efficiently handle their clinical documentation needs with confidence and ease.
How to create a PDF with pdfFiller
Who needs this?
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms