Finish Table in the Clinical Trial Agreement Template with ease For Free
Drop document here to upload
Up to 100 MB for PDF and up to 25 MB for DOC, DOCX, RTF, PPT, PPTX, JPEG, PNG, JFIF, XLS, XLSX or TXT
Note: Integration described on this webpage may temporarily not be available.
0
Forms filled
0
Forms signed
0
Forms sent
Discover the simplicity of processing PDFs online

Upload your document in seconds

Fill out, edit, or eSign your PDF hassle-free

Download, export, or share your edited file instantly
Top-rated PDF software recognized for its ease of use, powerful features, and impeccable support






Every PDF tool you need to get documents done paper-free
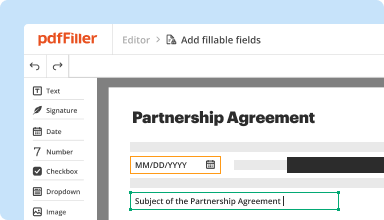
Create & edit PDFs
Generate new PDFs from scratch or transform existing documents into reusable templates. Type anywhere on a PDF, rewrite original PDF content, insert images or graphics, redact sensitive details, and highlight important information using an intuitive online editor.
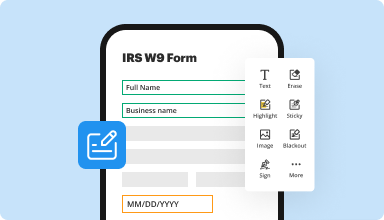
Fill out & sign PDF forms
Say goodbye to error-prone manual hassles. Complete any PDF document electronically – even while on the go. Pre-fill multiple PDFs simultaneously or extract responses from completed forms with ease.
Organize & convert PDFs
Add, remove, or rearrange pages inside your PDFs in seconds. Create new documents by merging or splitting PDFs. Instantly convert edited files to various formats when you download or export them.
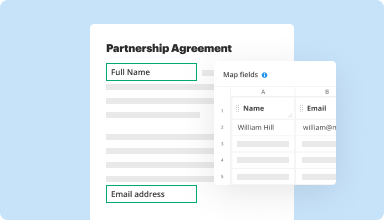
Collect data and approvals
Transform static documents into interactive fillable forms by dragging and dropping various types of fillable fields on your PDFs. Publish these forms on websites or share them via a direct link to capture data, collect signatures, and request payments.

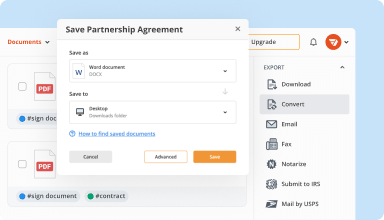
Export documents with ease
Share, email, print, fax, or download edited documents in just a few clicks. Quickly export and import documents from popular cloud storage services like Google Drive, Box, and Dropbox.
Store documents safely
Store an unlimited number of documents and templates securely in the cloud and access them from any location or device. Add an extra level of protection to documents by locking them with a password, placing them in encrypted folders, or requesting user authentication.
Customer trust by the numbers
64M+
users worldwide
4.6/5
average user rating
4M
PDFs edited per month
9 min
average to create and edit a PDF
Join 64+ million people using paperless workflows to drive productivity and cut costs
Why choose our PDF solution?
Cloud-native PDF editor
Access powerful PDF tools, as well as your documents and templates, from anywhere. No installation needed.
Top-rated for ease of use
Create, edit, and fill out PDF documents faster with an intuitive UI that only takes minutes to master.
Industry-leading customer service
Enjoy peace of mind with an award-winning customer support team always within reach.
What our customers say about pdfFiller
See for yourself by reading reviews on the most popular resources:
Although I initially had thought this was free, the overall experience is a positive one. The insertion process could be initially a little more self-setting / precise but overall good to use for my purposes
2016-07-13
It'd be helpful if there was a visual display on the side when selecting a document, so that I could see what I'm looking at and have the ability to rename it without completely opening it.
2017-08-20
I just was able to get the best service…
I just was able to get the best service ever from Paul. I found I did not use this service as much as I did in the past, but the automatic renewal of payment had gone through. I contacted CS via Support and received a FULL Refund and cancellation of Auto Pay with in 3 minutes flat. Furthermore, received an email with confirmation of my request!! This is the way to do business!! Thank you for making the refund process painless!
2019-05-16
What do you like best?
The quick uploading of large documents and the highlight tool.
What do you dislike?
I don’t dislike too muchghkkbccv. Vbbnnnnn
What problems are you solving with the product? What benefits have you realized?
I like to highlight certain documents for my clients.
2021-02-15
What do you like best?
Ability to edit pdf files and general ease of use
What do you dislike?
Limitations on how many files I can upload and the fact that it opens the files immediately, If I want to merge a newly uploaded files with previously saved files on my dashboard, it creates some unnecessary steps.
What problems are you solving with the product? What benefits have you realized?
Not really any problems - just a few areas the program feels clunky.
2020-08-18
What do you like best?
The ease of converting documents to pdf to fill out online.
What do you dislike?
Download to my documents is not always simple. However usually can manage.
Recommendations to others considering the product:
Do it. Wonderful tool for business.
What problems are you solving with the product? What benefits have you realized?
I receive documents often from vendors, customers and regulators that require downloading and filling out. PDFfiller makes this so simple. Download, send to pdfFiller and fill out (often sign), done.
2020-08-12
Easy to import edit
Easy to import edit- wished I had this a long time ago! I was desperately trying to find a way to send my Doctor forms. Scanner/Printer was knocked out from the storm.
2020-07-23
Adapting from working home, I needed to make forms fillable to get signatures and this service was exactly what I needed!
Even after I forgot to cancel my subscription after the free trial, their customer support was auper responsive and able to refund the autopayment.
Thank you!
2020-06-26
Had an issue with my subscription
Had an issue with my subscription cancellation, was rectified within less than an hour by Kim and very efficient customer service.
2020-04-30
Finish Table in Clinical Trial Agreement Template
The Finish Table feature in our Clinical Trial Agreement Template simplifies the management of your clinical trials. It provides a clear, organized space to mark the completion of critical milestones, ensuring that your team stays on track throughout the study.
Key Features
User-friendly interface for easy navigation
Clear documentation of completed tasks
Real-time collaboration for team members
Automated reminders for upcoming deadlines
Customizable sections to fit your trial needs
Potential Use Cases and Benefits
Streamlining the tracking of clinical trial phases
Enhancing team communication and collaboration
Reducing the risk of missed deadlines
Facilitating compliance with regulatory requirements
Improving project management efficiency
By using the Finish Table, you can easily visualize and manage all aspects of your clinical trial, which ultimately leads to fewer errors and smoother operations. This feature addresses your need for better organization and clarity in a complex process. With it, your team can focus on the important tasks at hand, driving your trial towards success.
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
What if I have more questions?
Contact Support
Who are the members of a clinical trial?
Depending on the funding available and complexity of the trial design, the team will usually include: Site Principal Investigator and Associate Investigators. Biostatistician. Study Coordinator/Research Nurse. Data Manager. Programmer. Clinical Trial Pharmacist, for clinical trials of an investigational medicinal product.
What is a CTA in a clinical trial?
Definition. A clinical trial agreement (CTA) or clinical study agreement (CSA) is a legally binding agreement that governs the conduct of a particular study and sets forth the obligations of each party to the agreement.
Who are the parties involved in a clinical trial?
Participant: A person who volunteers to take part in the clinical trial—such as yourself! Investigator: A researcher who helps conduct the clinical trial—such as a doctor. Study coordinator: A person who works with the investigator to manage the clinical trial—such as a nurse.
Who signs a clinical trial agreement?
Clinical Trial Agreements (CTAs) require signatures from both the trial sponsor and a University institutional official (IO) with signature authority delegated by the Board of Trustees of the Leland Stanford Junior University.
Who are the participants of a clinical trial?
Many different types of people take part in clinical trials. Some studies include healthy volunteers, while other studies include patient volunteers. Some studies include both healthy and patient volunteers . In addition, the NHLBI is committed to supporting clinical trials with diverse participants.
What are the components of a clinical trial agreement?
Clinical Trial Agreements typically involve a trial sponsor (a pharmaceutical, biotech, or medical device company), a research institution and a principal investigator. Separate agreements are negotiated in their entirety for each clinical trial.
Who are the parties to a clinical trial?
Sponsor: The person or group of people who supervise or fund the trial—such as a drug company. Participant: A person who volunteers to take part in the clinical trial—such as yourself! Investigator: A researcher who helps conduct the clinical trial—such as a doctor.
Who are the major players in clinical trials?
Who Makes Up a Clinical Research Team? Principal investigator (PI) Also called a primary investigator, this person oversees all aspects of a clinical research study. Study physicians. Research nurse. Study coordinator. Research pharmacists. Participants.
#1 usability according to G2
Try the PDF solution that respects your time.






