Generate Break Release For Free
Drop document here to upload
Up to 100 MB for PDF and up to 25 MB for DOC, DOCX, RTF, PPT, PPTX, JPEG, PNG, JFIF, XLS, XLSX or TXT
Note: Integration described on this webpage may temporarily not be available.
0
Forms filled
0
Forms signed
0
Forms sent
Discover the simplicity of processing PDFs online

Upload your document in seconds

Fill out, edit, or eSign your PDF hassle-free

Download, export, or share your edited file instantly
Top-rated PDF software recognized for its ease of use, powerful features, and impeccable support






Every PDF tool you need to get documents done paper-free
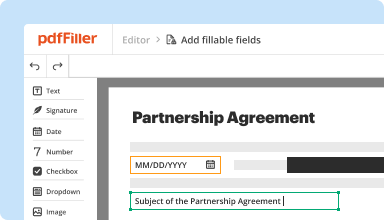
Create & edit PDFs
Generate new PDFs from scratch or transform existing documents into reusable templates. Type anywhere on a PDF, rewrite original PDF content, insert images or graphics, redact sensitive details, and highlight important information using an intuitive online editor.
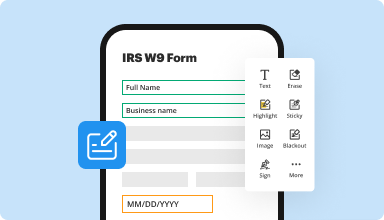
Fill out & sign PDF forms
Say goodbye to error-prone manual hassles. Complete any PDF document electronically – even while on the go. Pre-fill multiple PDFs simultaneously or extract responses from completed forms with ease.
Organize & convert PDFs
Add, remove, or rearrange pages inside your PDFs in seconds. Create new documents by merging or splitting PDFs. Instantly convert edited files to various formats when you download or export them.
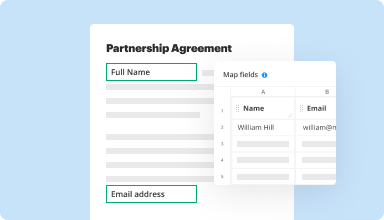
Collect data and approvals
Transform static documents into interactive fillable forms by dragging and dropping various types of fillable fields on your PDFs. Publish these forms on websites or share them via a direct link to capture data, collect signatures, and request payments.

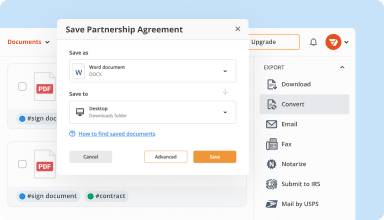
Export documents with ease
Share, email, print, fax, or download edited documents in just a few clicks. Quickly export and import documents from popular cloud storage services like Google Drive, Box, and Dropbox.
Store documents safely
Store an unlimited number of documents and templates securely in the cloud and access them from any location or device. Add an extra level of protection to documents by locking them with a password, placing them in encrypted folders, or requesting user authentication.
Customer trust by the numbers
64M+
users worldwide
4.6/5
average user rating
4M
PDFs edited per month
9 min
average to create and edit a PDF
Join 64+ million people using paperless workflows to drive productivity and cut costs
Why choose our PDF solution?
Cloud-native PDF editor
Access powerful PDF tools, as well as your documents and templates, from anywhere. No installation needed.
Top-rated for ease of use
Create, edit, and fill out PDF documents faster with an intuitive UI that only takes minutes to master.
Industry-leading customer service
Enjoy peace of mind with an award-winning customer support team always within reach.
What our customers say about pdfFiller
See for yourself by reading reviews on the most popular resources:
Only used twice but each time I was pleased with the features and ease of use. The only negative is that it is difficult to save files. I had to use a two step process of saving to PDF Filler then copying to my local drive.
2015-01-27
I am having trouble with the Schedule B when it gets half way down the page my screen jumps back to the top and I have a hard time making sure everything is on the right line
2018-11-08
What do you like best?
I can edit PDF files that I use often with ease.
What do you dislike?
I still can not figure out how to save as in the pdf filler to go back and edit the same exact file.
What problems are you solving with the product? What benefits have you realized?
I am saving time which is helping me save money.
I can edit PDF files that I use often with ease.
What do you dislike?
I still can not figure out how to save as in the pdf filler to go back and edit the same exact file.
What problems are you solving with the product? What benefits have you realized?
I am saving time which is helping me save money.
2019-01-29
What do you like best?
It gave us the opportunity to place our registration in a fillable format.
What do you dislike?
A user cannot save their progress and must complete the entire form in one sitting.
What problems are you solving with the product? What benefits have you realized?
Easy for parents to use. Parents no longer need to print out the document and send it in with a hard copy. It also gives us the ability to file a digital and hard copy if we so choose.
It gave us the opportunity to place our registration in a fillable format.
What do you dislike?
A user cannot save their progress and must complete the entire form in one sitting.
What problems are you solving with the product? What benefits have you realized?
Easy for parents to use. Parents no longer need to print out the document and send it in with a hard copy. It also gives us the ability to file a digital and hard copy if we so choose.
2019-05-28
It works well
Very easy to use and affordable, hasn't ever caused me an issue yet. I would recommend to anyone who needs to constantly convert files like I do
2023-10-25
Great Customer Service
Used the service on a free trial, was a positive experience but I didn't need it after I was done applying for apartments. Forgot to cancel after my trial and was charged for 2 months, contacted customer service on their live chat and was given the full refund within a minute! World class customer service!
2021-04-07
I needed this for school had everything that was neccesary
Nothing special in my opinion but it did what it was supposed to do without any problems so 5 star :D
2021-04-04
Great customer service and solid product.
I don't usually leave reviews, but this product deserves my time.
Works very well in filling PDFs. Typing, checking boxes (using the checkmark or "x" feature), filling out tables, etc. Saving and editing again works well, though it isn't entirely intuitive the first time.
Customer service was absolutely awesome! I realized, after signing up for a paid subscription, that I no longer needed the service. I cancelled online (or THOUGHT I did), but I must've had a glitch. When I got billed, I called and they issued me a refund no questions asked and zero hassle.
Customer service was incredibly helpful. Product was great for filling out multiple long pdfs that only had slight variations (saved a copy and changed only the fields that I needed). Easy to use and quick to learn.
I cancelled my subscription only because I realized I wouldn't be using it enough to justify the cost. Life changes!
2020-09-01
I am pleased with how easy it is to use…
I am pleased with how easy it is to use the app. The support given is great as any concerns I had were dealt with promptly and professionally.
2025-02-25
Generate Break Release Feature
The Generate Break Release feature simplifies your workflow by allowing you to quickly and efficiently manage breaks in your processes. This tool empowers you to maintain productivity while ensuring a smooth transition between tasks.
Key Features
Automated break detection to save time
Customizable settings for tailored use
Easy integration with existing systems
User-friendly interface for seamless navigation
Real-time notifications for updates
Potential Use Cases and Benefits
Improve efficiency in manufacturing processes
Streamline operations in customer service
Enhance project management in software development
Facilitate smoother transitions in logistics
Reduce downtime in various industries
With the Generate Break Release feature, you can solve common issues related to workflow interruptions. By automating break management, you reduce manual errors and increase efficiency. This results in higher productivity and less frustration, allowing you to focus on what truly matters—achieving your goals.
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
What if I have more questions?
Contact Support
Is energy released when bonds are broken or formed?
Energy is absorbed to break bonds. Bond-breaking is an endothermic process. Energy is released when new bonds form. Bond-making is an exothermic process.
Why is energy released when a bond is formed?
Energy is required to break bonds. Atoms are much happier when they are “married” and release energy because it is easier and more stable to be in a relationship (e.g., to generate octet electronic configurations). The enthalpy change is negative because the system is releasing energy when forming bond.
Is energy released when bonds are made?
Energy is absorbed to break bonds. ... Energy is released when new bonds form. Bond-making is an exothermic process. Whether a reaction is endothermic or exothermic depends on the difference between the energy needed to break bonds and the energy released when new bonds form.
Is heat released when bonds are formed?
Most chemical reactions involve the breaking and formation of chemical bonds. It takes energy to break a chemical bond, but energy is released when chemical bonds are formed. If more energy is released than consumed, then the chemical reaction evolves heat and is said to be exothermic.
Why is bond formation exothermic?
When bonds are formed the system loses energy and hence increases its stability (which is the ultimate motive). Since there is a decrease in energy, the energy lost is released as heat energy, and thus it is an exothermic process.
What happens to energy when a chemical bond is broken?
Chemical reactions make and break the chemical bonds between molecules, resulting in new materials as the products of the chemical reaction. ... Breaking chemical bonds absorbs energy, while making new bonds releases energy, with the overall chemical reaction being endothermic or exothermic.
When a chemical bond is broken energy is released or absorbed?
Energy is absorbed to break bonds. Bond-breaking is an endothermic process. Energy is released when new bonds form. Bond-making is an exothermic process.
Why does breaking a chemical bond always require energy?
Making chemical bonds always releases energy because the bond is formed in order to make the atoms more stable (less energy). ... Breaking chemical bonds always requires energy because the atoms are held together by the interactions of electrons in their valence shells. Separating them therefore makes them less stable.
What happens when chemical bond is formed?
When a chemical reaction occurs, molecular bonds are broken and other bonds are formed to make different molecules. For example, the bonds of two water molecules are broken to form hydrogen and oxygen. Energy is always required to break a bond, which is known as bond energy. ... Energy is released when a bond is made.
What energy changes occur when chemical bonds are formed and broken?
When a bod is broke, energy is required to break the bond, hence energy is taken in or absorbed when bonds are broken. This makes bond breaking an endothermic process. However, energy is released when bonds are made. This causes a net production of energy, which in turn makes the reaction exothermic in nature.
#1 usability according to G2
Try the PDF solution that respects your time.






