Rubber-stamp Initial Clinical Trial Agreement Template For Free




Join the world’s largest companies
How to Send a PDF for eSignature









Why choose pdfFiller for eSignature and PDF editing?

Cross-platform solution

Unlimited document storage

Widely recognized ease of use

Reusable templates & forms library
The benefits of electronic signatures

Efficiency

Accessibility

Cost savings

Security

Legality

Sustainability
Enjoy straightforward eSignature workflows without compromising data security

GDPR compliance

SOC 2 Type II Certified

PCI DSS certification

HIPAA compliance

CCPA compliance
Rubber-stamp Initial Clinical Trial Agreement Template
The Rubber-stamp Initial Clinical Trial Agreement Template simplifies the process of initiating clinical trials. This template provides a clear, concise framework that promotes efficiency while ensuring compliance with necessary regulations.
Key Features
Potential Use Cases and Benefits
This template addresses your need for a reliable and efficient way to manage initial clinical trial agreements. It eliminates guesswork, reduces paperwork, and fosters clear communication among all parties involved. With this resource, you can focus more on the trial itself, knowing you have a solid agreement in place.
Rubber-stamp Initial Clinical Trial Agreement Template with the swift ease
pdfFiller allows you to Rubber-stamp Initial Clinical Trial Agreement Template in no time. The editor's handy drag and drop interface ensures quick and intuitive signing on any device.
Ceritfying PDFs electronically is a quick and secure method to validate papers anytime and anywhere, even while on the fly.
See the step-by-step guide on how to Rubber-stamp Initial Clinical Trial Agreement Template electronically with pdfFiller:
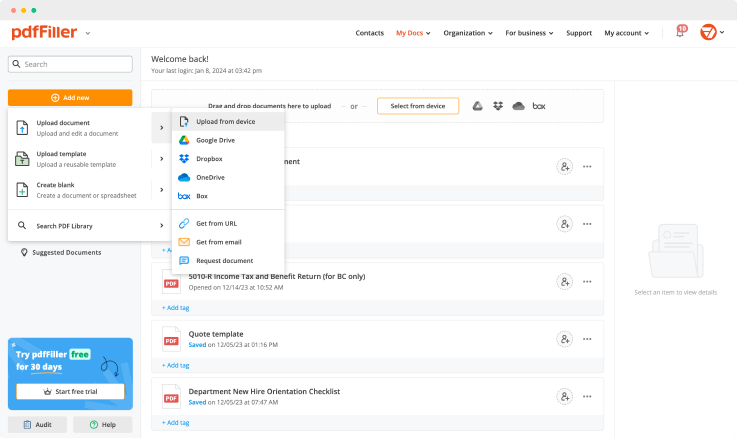
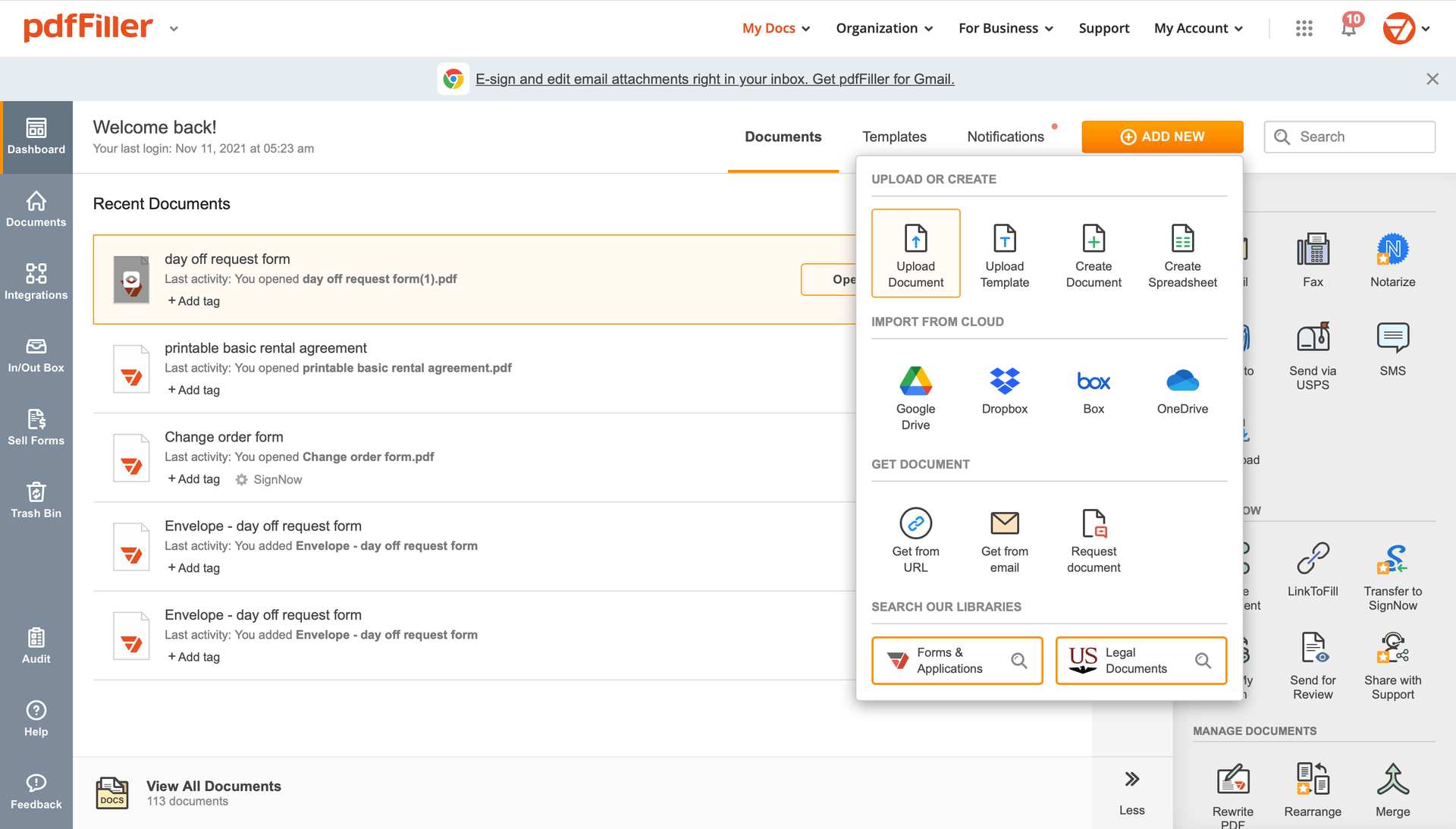
Add the form you need to sign to pdfFiller from your device or cloud storage.
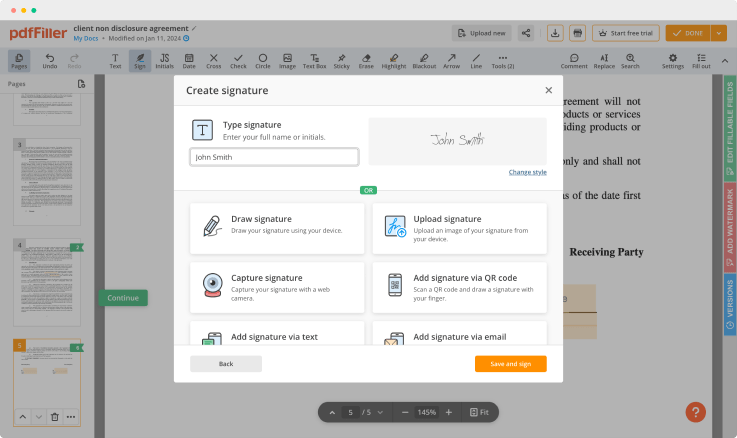
As soon as the document opens in the editor, click Sign in the top toolbar.

Create your electronic signature by typing, drawing, or importing your handwritten signature's image from your device. Then, hit Save and sign.
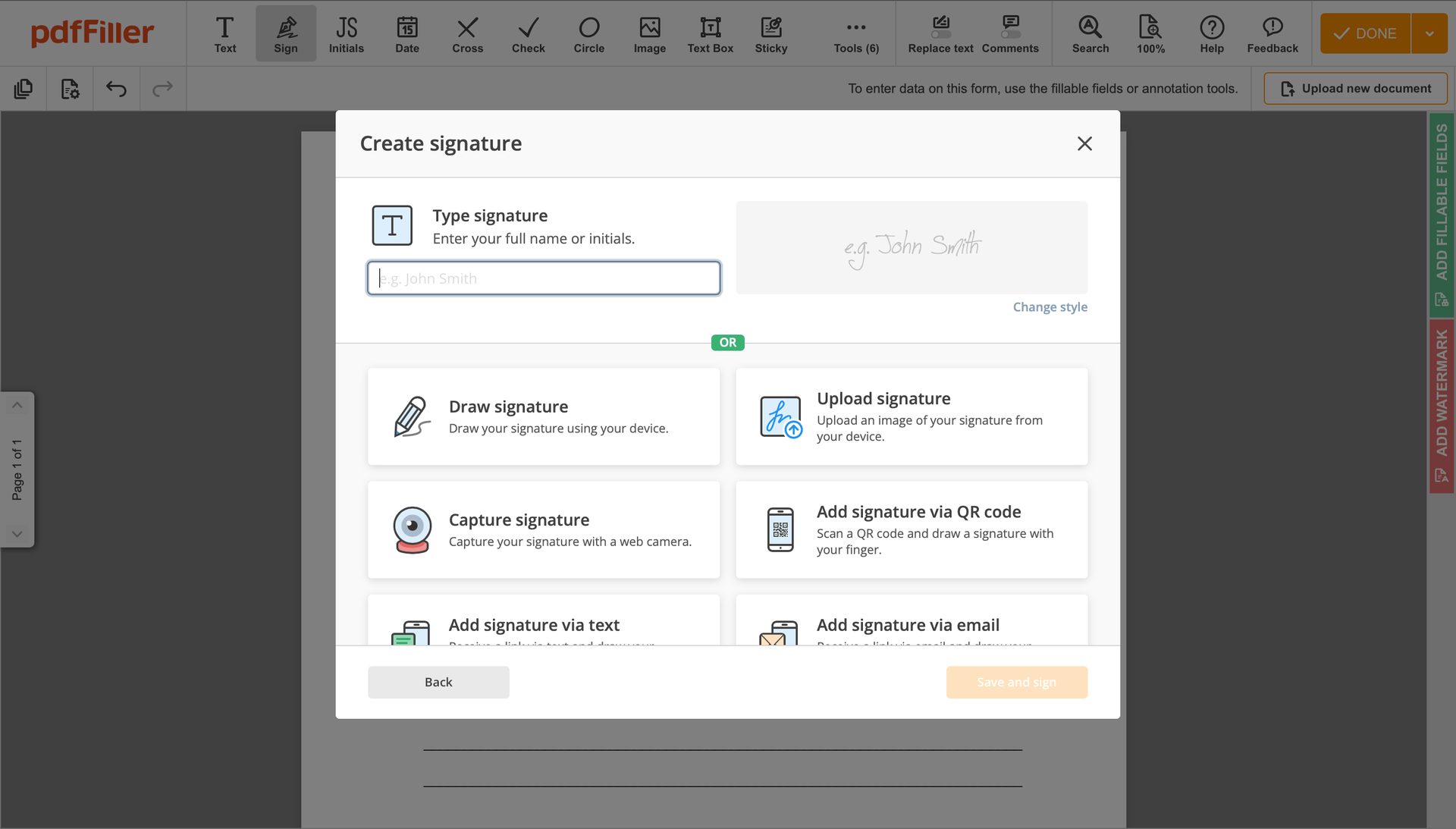
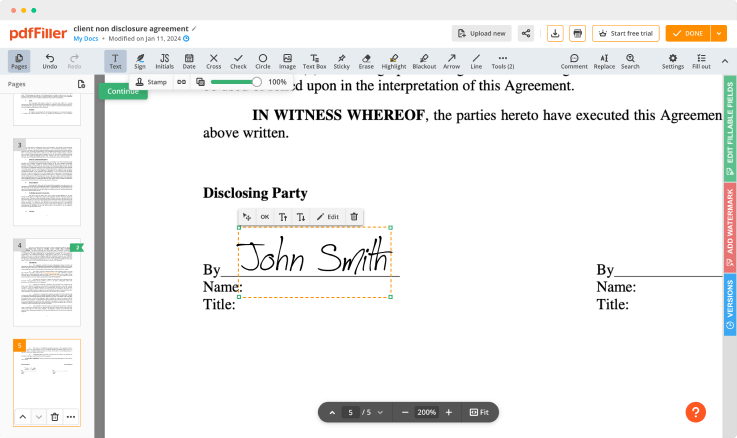
Click anywhere on a document to Rubber-stamp Initial Clinical Trial Agreement Template. You can drag it around or resize it using the controls in the floating panel. To use your signature, hit OK.
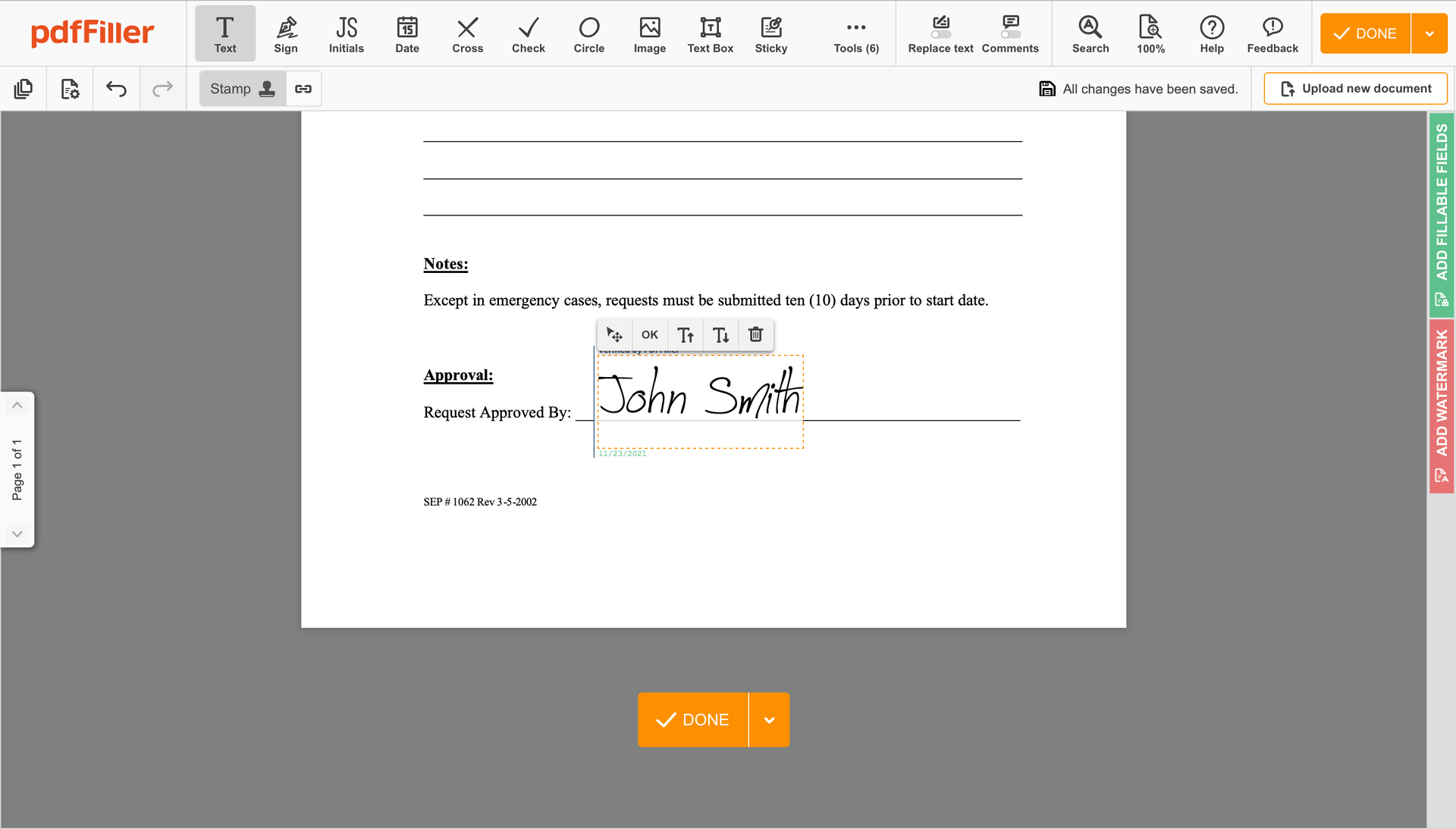
Finish up the signing session by clicking DONE below your document or in the top right corner.
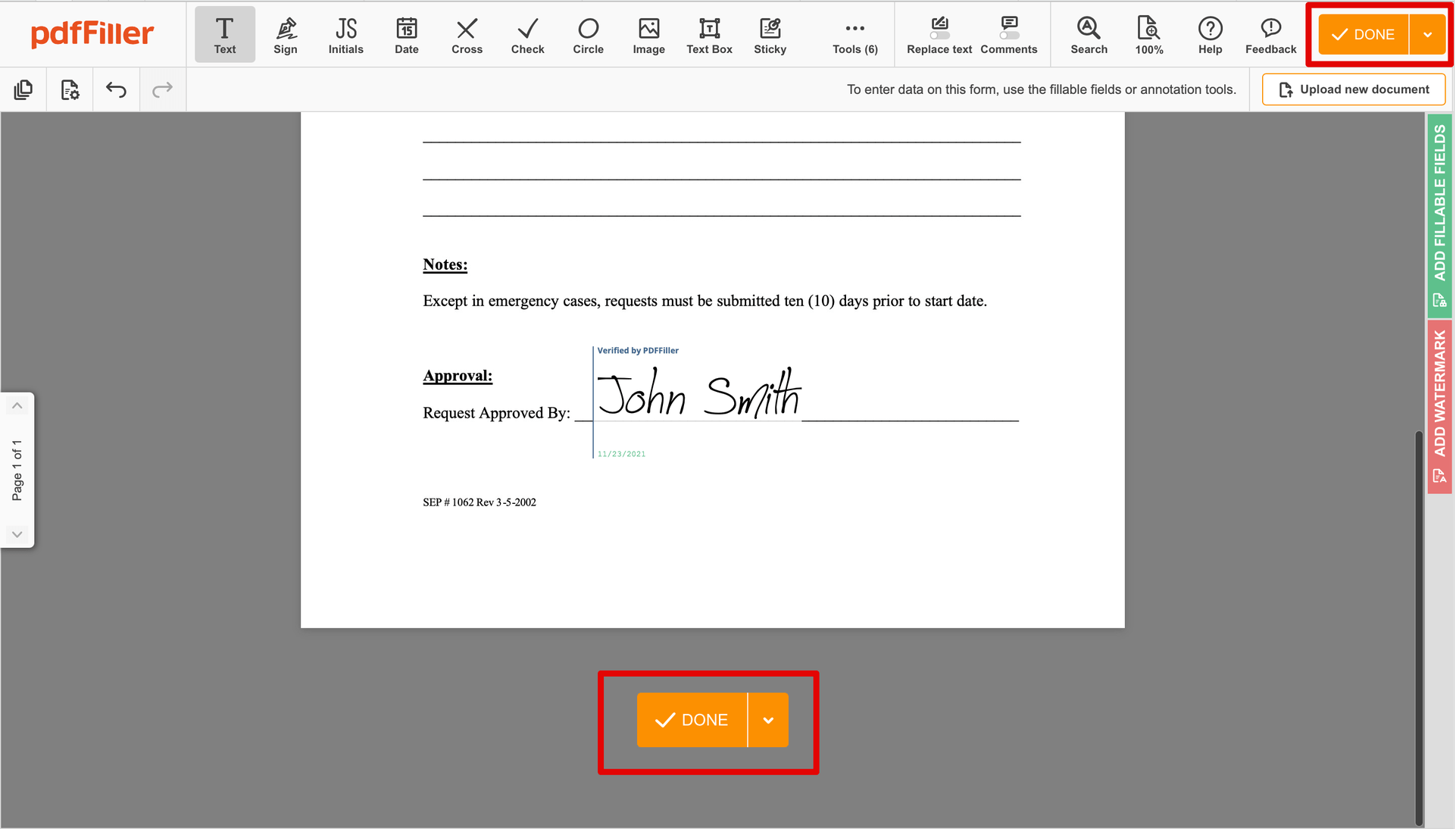
Next, you'll return to the pdfFiller dashboard. From there, you can get a completed copy, print the form, or send it to other parties for review or validation.
Are you stuck with different applications for managing documents? Use our solution instead. Use our tool to make the process efficient. Create document templates completely from scratch, edit existing forms, integrate cloud services and utilize even more useful features without leaving your account. You can Rubber-stamp Initial Clinical Trial Agreement Template right away, all features are available instantly. Pay as for a lightweight basic app, get the features as of pro document management tools.
How to edit a PDF document using the pdfFiller editor:
Ready to try pdfFiller's? Rubber-stamp Initial Clinical Trial Agreement Template