Secure legal documents with Confidential Disclosure In Clinical Research Agreement Template creator tool
Secure legal documents with Confidential Disclosure In Clinical Research Agreement Template creator tool with pdfFiller
What is a Confidential Disclosure In Clinical Research Agreement?
A Confidential Disclosure In Clinical Research Agreement (CDCRA) is a legal contract that protects sensitive information shared among parties involved in clinical research. It ensures that proprietary data, processes, and methodologies remain confidential and are not disclosed without permission. Such agreements are crucial in medical, pharmaceutical, and biotechnology fields where intellectual property and trade secrets are at stake.
Why organizations use a Confidential Disclosure In Clinical Research Agreement?
Organizations leverage CDCRAs to safeguard their research data, maintain competitive advantage, and comply with regulatory requirements. By formalizing confidentiality, they can foster collaboration while minimizing the risks of data leakage or intellectual property theft. This practice is increasingly important in an era where data integrity and security are paramount.
Core functionality of the Confidential Disclosure In Clinical Research Agreement creator tool in pdfFiller
The Confidential Disclosure In Clinical Research Agreement creator tool in pdfFiller enables users to efficiently create secure legal documents. This tool features easy-to-navigate templates, real-time editing, collaborative capabilities, and automated eSigning. With a cloud-based architecture, users can access and edit their documents from anywhere, ensuring their confidentiality and compliance throughout the document lifecycle.
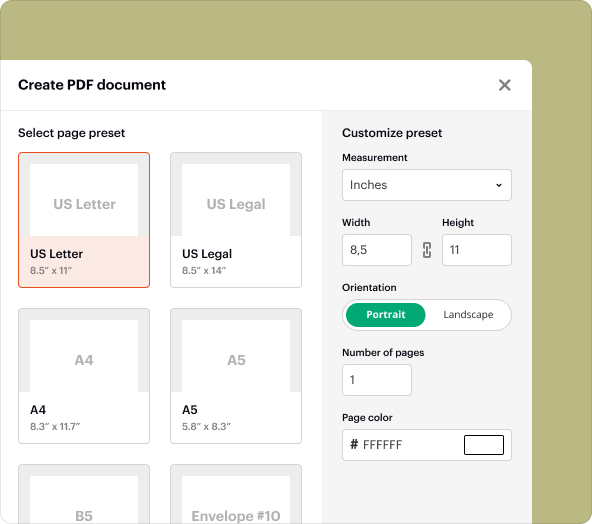
Step-by-step: using the Confidential Disclosure In Clinical Research Agreement creator tool to create blank PDFs
To create a blank PDF using pdfFiller's CDCRA tool, follow these steps:
-
Log into your pdfFiller account.
-
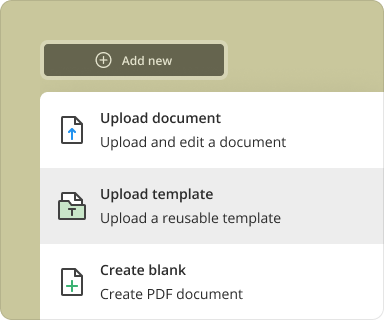
Select the 'Create' option and choose 'Blank Document.'
-
Access the 'Templates' section and select 'Confidential Disclosure In Clinical Research Agreement.'
-
Fill in the relevant fields with your specific details.
-
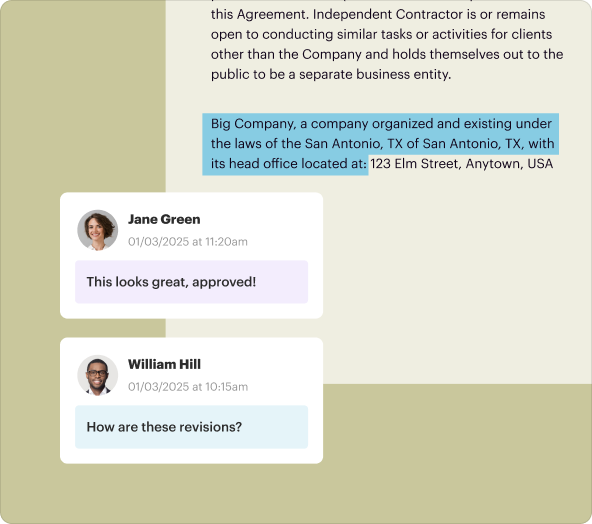
Review the document and make any necessary edits before saving.
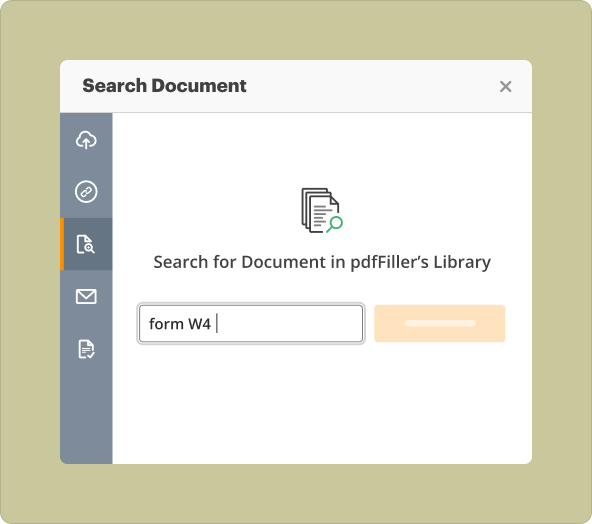
Creating new PDFs from scratch vs starting with existing files in the creator tool
Users can either start from a blank PDF or upload an existing document. Creating from scratch allows for complete customization, while uploading an existing file provides a foundation that can be modified as required. Both methods have advantages: starting fresh gives more flexibility, while utilizing an existing document can save time.
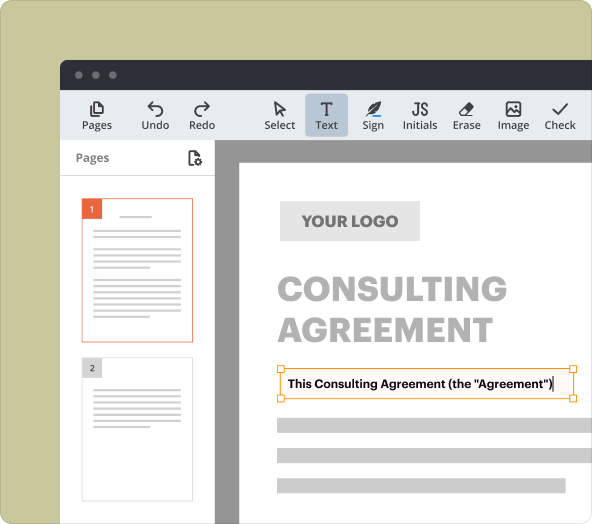
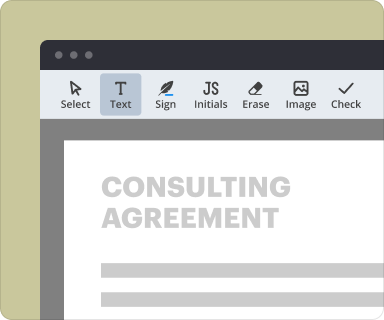
Structuring and formatting text within PDFs via the creator tool
Formatting text in your Confidential Disclosure In Clinical Research Agreement is straightforward with pdfFiller. You can adjust text alignment, font size, and style to ensure clarity and professionalism. Adding tables or bullet points can further enhance the document's readability, which is vital for legal documents.
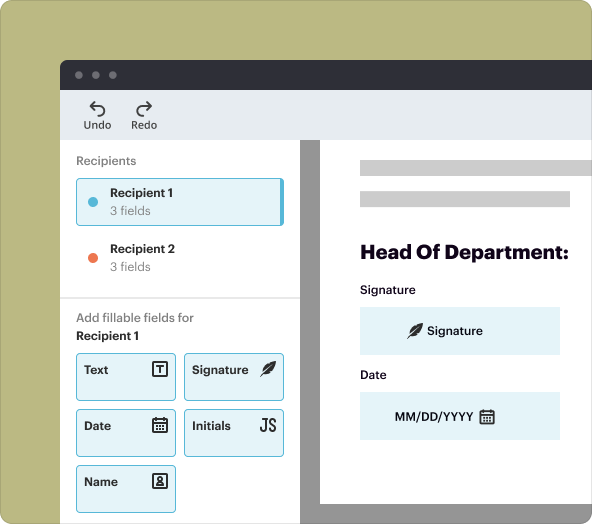
Saving, exporting, and sharing documents made with the creator tool
Once your CDCRA is finalized, pdfFiller allows for easy saving and sharing. You can export your document in various formats (such as PDF, Word, or Excel) or share it directly via email. The platform also supports secure storage, ensuring that your sensitive agreements remain protected.
Typical industries and workflows that depend on the creator tool
The Confidential Disclosure In Clinical Research Agreement creator tool is utilized primarily within the healthcare, pharmaceutical, and research sectors. These industries frequently require secure agreements to protect their research findings and proprietary information. Workflows often involve collaborative projects among clinical research organizations, ethics boards, and partner companies.
Conclusion
Securing legal documents with a Confidential Disclosure In Clinical Research Agreement Template creator tool through pdfFiller streamlines the process of drafting and managing essential agreements. With its robust features, ease of use, and collaborative capabilities, pdfFiller offers an essential solution for individuals and teams looking to protect their confidential data efficiently. Ensuring confidentiality in clinical research is critical, and leveraging the right tools is essential for compliance and security.
How to create a PDF with pdfFiller
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms




Ability to create templates (e.g. for billing) and changing dates when needed.
What do you dislike?
too much to choose from when importing records from the library
What problems are you solving with the product? What benefits have you realized?
Able to access files and email or fax at will when on the road.