Secure legal documents with Investigator Initiated Clinical Trial Agreement Template builder tool
Secure legal documents with Investigator Initiated Clinical Trial Agreement Template builder tool with pdfFiller
How to secure legal documents with Investigator Initiated Clinical Trial Agreement Template builder tool
To secure legal documents using the Investigator Initiated Clinical Trial Agreement Template builder tool in pdfFiller, simply start by selecting the template and customizing it to fit your needs. You can edit the text, add or remove sections, and use the e-signature feature to validate the agreement. Finally, save your document securely on the cloud or export it in your desired format.
What is an Investigator Initiated Clinical Trial Agreement?
An Investigator Initiated Clinical Trial Agreement (IITCA) is a formal legal document between a research investigator and an organization that outlines the terms under which a clinical trial will be conducted. This agreement typically includes aspects such as study objectives, funding responsibilities, patient recruitment strategies, data management protocols, and intellectual property rights. Securing such documents is critical to ensure compliance with legal and regulatory standards.
Why organizations use an Investigator Initiated Clinical Trial Agreement
Organizations often utilize IITCAs to protect intellectual property, establish funding outlines, and clarify roles and responsibilities. These agreements also ensure compliance with regulatory standards, enhance collaborative research efforts, and streamline communication among parties involved in the clinical trial. By securing these legal documents, organizations can reduce the risk of disputes and enhance the integrity of their research initiatives.
Core functionality of the Investigator Initiated Clinical Trial Agreement in pdfFiller
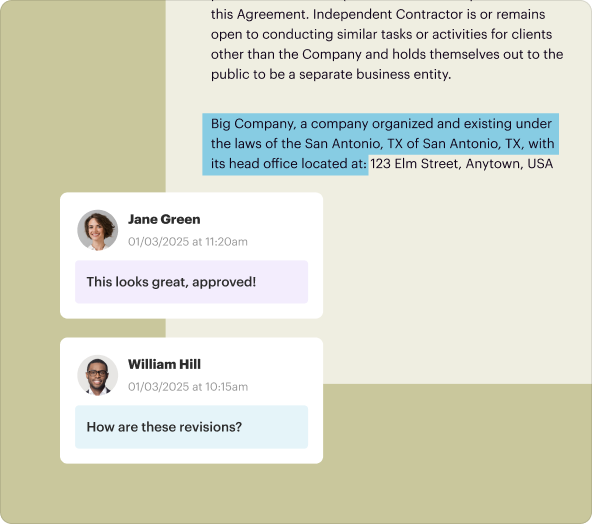
The Investigator Initiated Clinical Trial Agreement template builder tool in pdfFiller empowers users to create and customize their agreements efficiently. Key functionalities include versatile editing capabilities, e-signature integration, cloud storage for easy access, and sharing options for collaborative feedback. Users can also collaborate in real-time, making it an excellent tool for teams working on complex trials.
Step-by-step: using the Investigator Initiated Clinical Trial Agreement to create blank PDFs
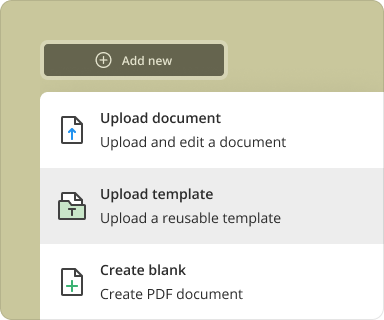
Creating a blank PDF with the Investigator Initiated Clinical Trial Agreement template in pdfFiller is straightforward. Follow these steps:
-
Log in to your pdfFiller account or create one if you’re a new user.
-
Navigate to the template library and search for 'Investigator Initiated Clinical Trial Agreement'.
-
Select the template and click on 'Use this Template'.
-
Customize the fields according to your clinical trial requirements.
-
Add or delete sections as needed, then save your document.
Creating new PDFs from scratch vs starting with existing files in the Investigator Initiated Clinical Trial Agreement
Users can choose to create an Investigator Initiated Clinical Trial Agreement document from scratch or edit an existing document. Creating from scratch allows for complete customization from the start, tailoring each section to the exact needs of the study. Conversely, starting with an existing file can save time by providing a foundational structure that only requires modifications to align with specific trial requirements.
-
Complete control over the document content and structure.
-
May require more initial time investment.
-
Faster to deploy as the base structure is already present.
-
Less control over pre-existing content that may need extensive editing.
Structuring and formatting text within PDFs via the Investigator Initiated Clinical Trial Agreement
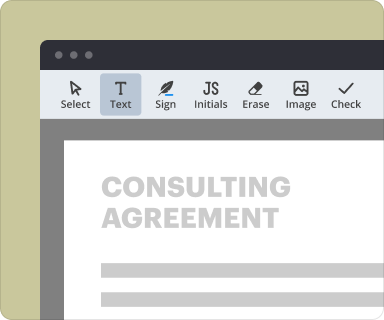
pdfFiller provides robust tools for structuring and formatting text within your Investigator Initiated Clinical Trial Agreement. Users can adjust font sizes, styles, and paragraph alignments to ensure the document is professional and compliant. Additionally, inserting forms, tables, or images is simple, enabling the creation of a comprehensive and visually appealing document.
Saving, exporting, and sharing documents made with the Investigator Initiated Clinical Trial Agreement
Once your Investigator Initiated Clinical Trial Agreement is finalized, pdfFiller offers various options for saving and exporting documents. You can store your documents securely in the cloud, export them as PDF, Word, or JPEG formats, and share them easily with stakeholders through email or direct links. The ability to manage document versions and track changes ensures that you always have the latest agreement.
Typical industries and workflows that depend on the Investigator Initiated Clinical Trial Agreement
The healthcare and pharmaceuticals industries predominantly rely on the Investigator Initiated Clinical Trial Agreement. Research institutions, hospitals, and pharmaceutical companies utilize this document to facilitate clinical trials effectively. Teams engaged in research and development, regulatory compliance, and clinical operations often engage with this type of agreement, making it essential for their workflows.
-
Utilize agreements to streamline clinical trials and protect patient data.
-
Engage in IITCAs to outline funding responsibilities and intellectual property rights.
-
Implement IITCAs to ensure collaborative research efforts are legally sound.
Conclusion
In conclusion, securing legal documents with the Investigator Initiated Clinical Trial Agreement Template builder tool in pdfFiller provides a streamlined, efficient approach to managing critical trial agreements. With its user-friendly interface and robust capabilities, pdfFiller simplifies the complexities of document creation, allowing organizations to focus on what matters most: conducting successful clinical trials while ensuring compliance and regulatory adherence.
How to create a PDF with pdfFiller
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms




I enjoy the ease of being able to upload a PDF file and edit the file online and add or delete content as necessary. I then download the completed PDF file to my documents.
What do you dislike?
I'm not sure this is a dislike but if I send a completed PDF document with a requested signature to a client the client Gets charged a fee.
Recommendations to others considering the product:
Pdffiller is a great way to complete additional documents are contracts used on a daily basis
What problems are you solving with the product? What benefits have you realized?
The ease of creating a document helps make business go faster. Duplicating documents that require only minor changes.