Secure legal documents with Investigator Initiated Clinical Trial Agreement Template creator software
Secure legal documents with Investigator Initiated Clinical Trial Agreement Template creator software
How to secure legal documents with Investigator Initiated Clinical Trial Agreement Template creator software
To securely handle legal documents such as Investigator Initiated Clinical Trial Agreements, utilize pdfFiller to create and manage your PDFs effectively. The platform allows you to create, edit, eSign, and share documents with ease, ensuring secure and compliant management of your critical information.
What is an Investigator Initiated Clinical Trial Agreement?
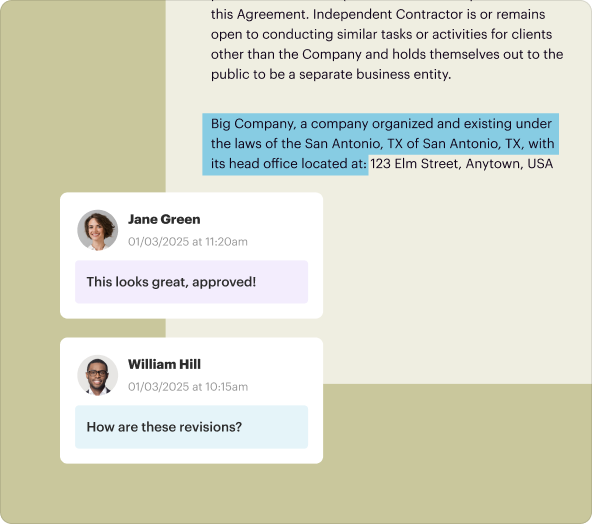
An Investigator Initiated Clinical Trial Agreement (IICCTA) is a legally binding document that outlines essential terms, roles, and responsibilities between parties participating in a clinical trial. It is especially significant for trials initiated by non-commercial sponsors, often academic institutions or research organizations. Clear delineation in these agreements helps mitigate risks and ensures regulatory compliance.
Why organizations use a PDF creator for Investigator Initiated Clinical Trial Agreements
Organizations rely on PDF creator software to streamline the documentation process of IICCTAs due to their security, versatility, and ease of use. By using such software, users can ensure that sensitive data remains confidential while still allowing for easy access and distribution among stakeholders.
Core functionality of pdfFiller for creating Investigator Initiated Clinical Trial Agreements
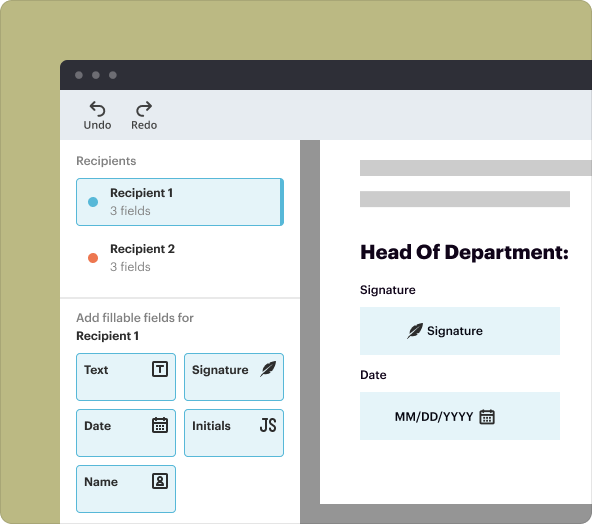
pdfFiller enables users to effectively create secure legal documents through a variety of integrated features. These include advanced editing tools, eSigning capabilities, collaboration mechanisms, and document templates specifically designed for legal agreements.
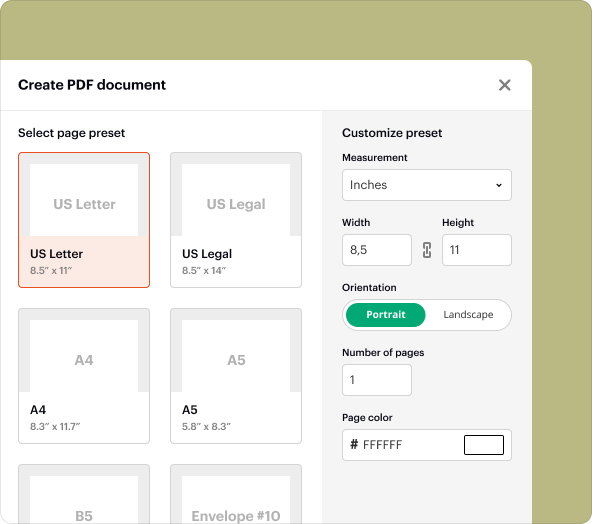
Step-by-step: using pdfFiller to create blank PDFs
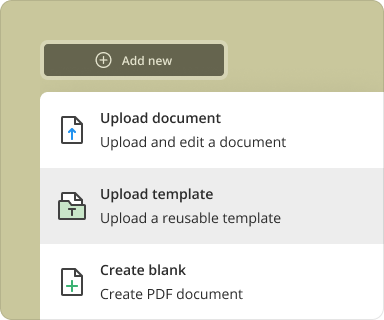
Creating a blank PDF using pdfFiller is straightforward. Follow these steps:
-
Log in to pdfFiller and click on 'Create New Document'.
-
Select 'Blank Document' or choose one of the templates available.
-
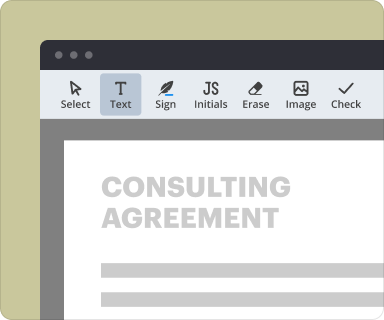
Utilize the editing tools to add text, checkboxes, or other elements to your document.
-
Save your document and opt for eSigning if necessary.
Creating new PDFs from scratch vs starting with existing files in pdfFiller
Choosing to create a PDF from scratch can be beneficial for complete customization. However, starting with an existing template or document for IICCTAs can ensure adherence to legal standards and save time. Compare the advantages:
-
Complete customization to meet specific needs.
-
Requires more time and legal knowledge.
-
Faster to complete; templates reduce the risk of missing key legal requirements.
-
May limit creative control or not fully address unique requirements.
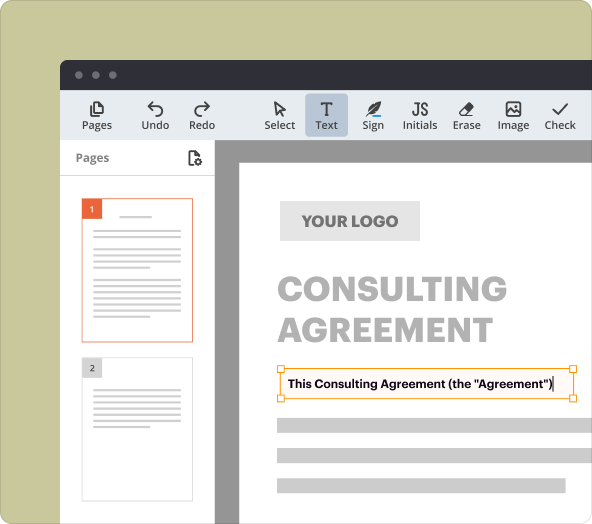
Organizing content and formatting text as you create IICCTAs
Proper organization of content within your legal documents is crucial. pdfFiller provides robust formatting options that allow you to structure your agreement clearly, ensuring all necessary sections are well-defined and easy to navigate.
Saving, exporting, and sharing documents made with pdfFiller
Once you've created your IICCTA, saving, exporting, and sharing become equally important. pdfFiller allows for seamless saving in various formats, including PDF and DOCX. Users can also share documents directly via email or through a secure link.
Typical industries and workflows that depend on PDF creators for IICCTAs
Various industries, including pharmaceuticals, academic institutions, and clinical research organizations, extensively use IICCTAs. The workflows generally involve collaboration among researchers, sponsors, and regulatory bodies, highlighting the need for robust document management solutions.
Conclusion
Using pdfFiller for creating and managing Investigator Initiated Clinical Trial Agreements streamlines the intricate process of legal documentation. With its comprehensive features and user-friendly interface, pdfFiller empowers individuals and teams to securely create legal documents efficiently, making it an invaluable tool in the clinical trial landscape.
How to create a PDF with pdfFiller
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms




How simple it is to use. I can sign on anywhere and make edits to pdf's. I can also convert documents. I've been using pdf filler for almost 3 years now and absolutely love it! Just renewed my subscription for year 4!
What do you dislike?
No complaints. Even on the rare occasion where the system crashes, the pdf filler team sends you an email to let you know they're working on correcting the problem. It's usually fixed within minutes.
Recommendations to others considering the product:
Don't think about it twice, use it!
What problems are you solving with the product? What benefits have you realized?
Last minute edits. Also love the signature feature that includes the dates. That comes in very handy in my field, which is real estate.