How to Pharmacovigilance Audit Checklist Template with pdfFiller
Learn how to generate a Pharmacovigilance Audit Checklist Template using pdfFiller, a cloud-based document creation platform that makes it easy to edit, share, and manage PDFs tailored to your needs.
What is a Pharmacovigilance Audit Checklist Template?
A Pharmacovigilance Audit Checklist Template is a structured document that assists organizations in systematically reviewing and assessing their pharmacovigilance practices. It typically contains a series of questions or criteria that help evaluate compliance with regulations and standards related to drug safety monitoring. These checklists are essential tools for ensuring thorough audits and fostering accountability in safety data management.
Why you might need a Pharmacovigilance Audit Checklist Template?
Organizations engaged in drug development and monitoring require a Pharmacovigilance Audit Checklist Template to ensure compliance with regulatory requirements and to uphold the highest safety standards. Such templates facilitate:
-
1.Consistency in audits across different departments and teams.
-
2.Identification of gaps in current practices and protocols.
-
3.Streamlined training for new staff by providing clear guidelines.
-
4.Documentation for regulatory inspections and assessments.
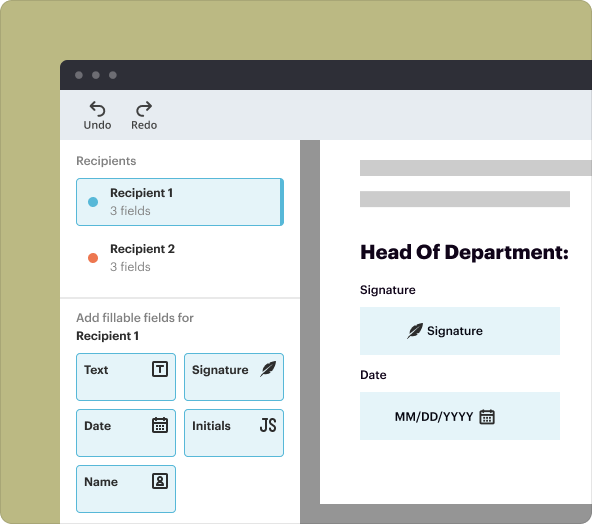
Key tools in pdfFiller that let you create a Pharmacovigilance Audit Checklist Template
pdfFiller offers a variety of powerful features that make the creation of a Pharmacovigilance Audit Checklist Template straightforward:
-
1.**User-friendly interface:** Easy navigation and drag-and-drop functionality.
-
2.**Template library:** Access to pre-existing templates for quick customization.
-
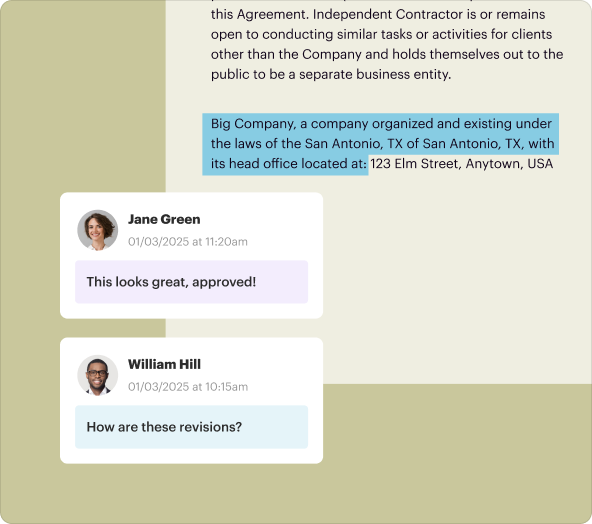
3.**Collaboration tools:** Real-time editing and comments to facilitate teamwork.
-
4.**E-signature capability:** Secure sign-off from relevant stakeholders.
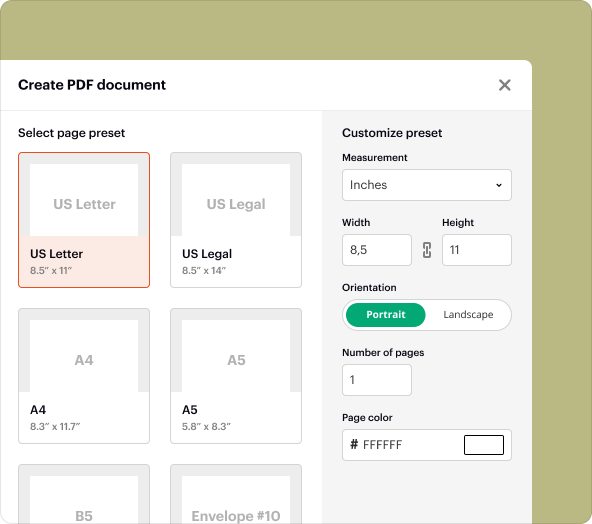
Step-by-step guide to creating blank PDFs for your Pharmacovigilance Audit Checklist Template
Creating a Pharmacovigilance Audit Checklist Template from scratch in pdfFiller is easy. Follow these steps:
-
1.Log in to your pdfFiller account.
-
2.Select 'Create New' and choose 'Blank Document' from the dropdown menu.
-
3.Use the editing tools to add text fields, checkboxes, and sections as per your audit criteria.
-
4.Save your document as a PDF once you have entered all necessary content.
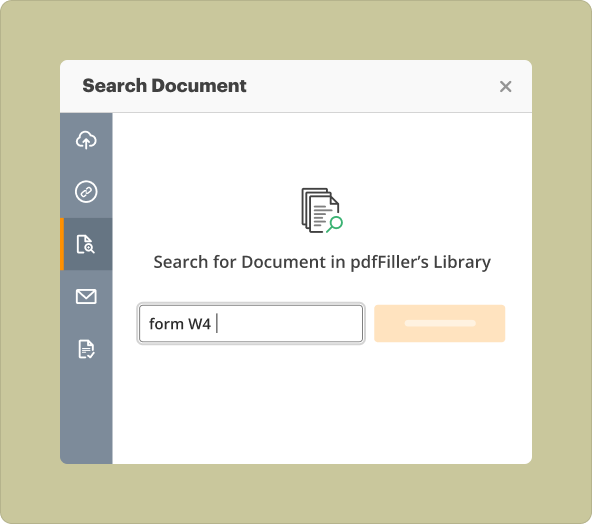
Pharmacovigilance Audit Checklist Template from scratch vs uploading existing files to modify
When deciding to create a Pharmacovigilance Audit Checklist Template, you have two primary options:
-
1.**Creating from scratch:** This allows for complete customization, enabling you to tailor the checklist to your specific requirements.
-
2.**Uploading existing files:** This option is beneficial when you have a pre-used template that requires updates. Simply upload it to pdfFiller, edit as needed, and save.
Organizing content and formatting text as you create your Pharmacovigilance Audit Checklist Template
Content organization and formatting are key elements in creating an effective checklist. Here are some tips for structuring your document:
-
1.**Use clear headings:** These guide users through different sections of the checklist.
-
2.**Employ bullet points or lists:** These make the checklist easier to scan and complete.
-
3.**Highlight essential criteria:** Use bold text or colors to draw attention to critical sections.
Saving, exporting, and sharing once you create your Pharmacovigilance Audit Checklist Template
Once your Pharmacovigilance Audit Checklist Template is complete, pdfFiller allows you to save, export, and share your PDF easily:
-
1.**Saving:** Save your document directly to the pdfFiller cloud for quick access.
-
2.**Exporting:** Download the document in various formats, including PDF, Word, or Excel as needed.
-
3.**Sharing:** Collaborate with team members by sharing the document link or inviting them to edit.
Typical use-cases and sectors that often use a Pharmacovigilance Audit Checklist Template
Various sectors benefit from utilizing a Pharmacovigilance Audit Checklist Template, including:
-
1.**Pharmaceutical companies:** Ensuring compliance with health regulations.
-
2.**Clinical research organizations:** Streamlining audit processes and reporting.
-
3.**Healthcare providers:** Monitoring drug safety practices and reporting requirements.
-
4.**Regulatory agencies:** Conducting effective assessments and inspections.
Conclusion
In summary, leveraging pdfFiller to create a Pharmacovigilance Audit Checklist Template simplifies the audit process while ensuring regulatory compliance and safety. The combination of user-friendly tools, collaborative features, and cloud-based access makes pdfFiller an invaluable asset for individuals and teams involved in pharmacovigilance. Start creating your template today and see how pdfFiller can elevate your documentation capabilities.
How to create a PDF with pdfFiller
Who needs this?
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms