
Get the free FDA Inspection Report - fda
Show details
This document outlines the observations made by the FDA during an inspection of the Estrella Family Creamery, focusing on food safety compliance and sanitation practices.
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign fda inspection report
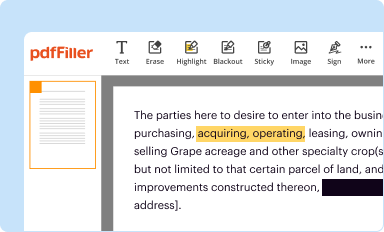
Edit your fda inspection report form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
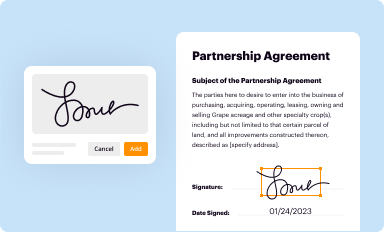
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your fda inspection report form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing fda inspection report online
Use the instructions below to start using our professional PDF editor:
1
Log in to account. Click on Start Free Trial and register a profile if you don't have one.
2
Prepare a file. Use the Add New button to start a new project. Then, using your device, upload your file to the system by importing it from internal mail, the cloud, or adding its URL.
3
Edit fda inspection report. Add and replace text, insert new objects, rearrange pages, add watermarks and page numbers, and more. Click Done when you are finished editing and go to the Documents tab to merge, split, lock or unlock the file.
4
Get your file. When you find your file in the docs list, click on its name and choose how you want to save it. To get the PDF, you can save it, send an email with it, or move it to the cloud.
pdfFiller makes working with documents easier than you could ever imagine. Register for an account and see for yourself!
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out fda inspection report
How to fill out FDA Inspection Report
01
Gather necessary information: Collect details about the facility, product, and inspection date.
02
Complete the header section: Fill in the name of the establishment, address, and other contact details.
03
Describe the type of inspection: Specify whether it's a routine inspection, for-cause inspection, etc.
04
Detail the purpose of the inspection: Clearly state the objectives of the inspection.
05
List observations: Note any findings, including compliance issues and areas of concern.
06
Document corrective actions: Indicate what actions are recommended or required to address issues.
07
Sign and date the report: Ensure the report is signed by the inspector and noted with the date of completion.
Who needs FDA Inspection Report?
01
Food and beverage manufacturers requiring compliance with safety regulations.
02
Pharmaceutical companies wishing to validate manufacturing processes.
03
Medical device manufacturers needing to demonstrate compliance.
04
Importers of food and drugs to ensure products meet U.S. safety standards.
05
Distributors and suppliers aiming for quality assurance in FDA regulated products.
Fill
form
: Try Risk Free






People Also Ask about
Are FDA inspection reports public?
The OII Electronic Reading Room displays copies of select foreign and domestic inspection and related records. We are making these records publicly available either (1) proactively at our discretion or (2) because they are "frequently requested" per the Electronic Freedom of Information Act Amendments of 1996.
Are FDA establishment inspection reports public?
Are FDA Inspection Reports Public? Yes, FDA inspection reports such as Establishment Inspection Reports (EIRs) can be requested through the Freedom of Information Act (FOIA). However, the process may take time, and certain proprietary information may be redacted.
Are FDA reports public?
The OII Electronic Reading Room displays copies of select foreign and domestic inspection and related records. We are making these records publicly available either (1) proactively at our discretion or (2) because they are "frequently requested" per the Electronic Freedom of Information Act Amendments of 1996.
Are FDA 483 observations public?
If you want to see any unpublished 483, you have to make a special request, which takes time and money. Theoretically, Form FDA 483s are public information and, thus, are available through the FDA's Freedom of Information Act Office. So, any Form FDA 483 can be requested by anyone.
Are FDA reports anonymous?
The FDA will not share your identity or contact information with anyone outside the FDA unless required to do so by law, regulation, or court order.
Where can I find FDA 483 reports?
What are the most common FDA 483 observations? Inadequate or insufficient testing of products or raw materials. Failure to properly handle, store, or label materials. Failure to establish or maintain adequate records or logs. Failure to properly report adverse events or incidents.
What are the 4 types of FDA inspections?
The four different types of FDA inspections are pre-approval inspection, routine inspection, compliance follow-up inspection, and “for cause” inspection. Each is intended to help protect the public from unsafe products, but the focus and expectations of each type of inspection are different.
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
What is FDA Inspection Report?
The FDA Inspection Report is a document that outlines the findings and observations made by the Food and Drug Administration during an inspection of a facility that produces food, drugs, medical devices, or other regulated products.
Who is required to file FDA Inspection Report?
Companies and facilities that are subject to FDA inspections are required to file an FDA Inspection Report, including manufacturers of food, pharmaceuticals, medical devices, and biologics.
How to fill out FDA Inspection Report?
To fill out the FDA Inspection Report, the inspector documents observations, findings, and any violations noted during the inspection, providing details such as date, facility information, and specific issues identified.
What is the purpose of FDA Inspection Report?
The purpose of the FDA Inspection Report is to assess compliance with federal regulations, ensure public safety, and document any deficiencies or concerns in product manufacturing processes.
What information must be reported on FDA Inspection Report?
The FDA Inspection Report must include information such as facility name and address, inspection date, observations made, specific regulatory violations, and recommendations for corrective actions.
Fill out your fda inspection report online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Fda Inspection Report is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.





















