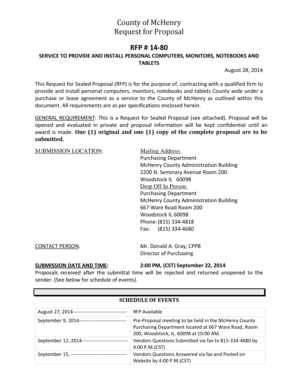
Get the free ) Describe the orbitals using in forming the triple bond in 2butyne
Show details
Name PHYS 0201 (Organic) Test 4 Fall 2004 1. (8 pts. Ea.) Describe the orbitals using in forming the triple bond in 2butyne; that is, H3C C 1. CH3 a. What is the hybridization of the two carbons involved
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign describe form orbitals using

Edit your describe form orbitals using form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.

Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your describe form orbitals using form via URL. You can also download, print, or export forms to your preferred cloud storage service.
How to edit describe form orbitals using online
Follow the steps below to benefit from a competent PDF editor:
1
Check your account. If you don't have a profile yet, click Start Free Trial and sign up for one.
2
Prepare a file. Use the Add New button to start a new project. Then, using your device, upload your file to the system by importing it from internal mail, the cloud, or adding its URL.
3
Edit describe form orbitals using. Add and replace text, insert new objects, rearrange pages, add watermarks and page numbers, and more. Click Done when you are finished editing and go to the Documents tab to merge, split, lock or unlock the file.
4
Get your file. Select your file from the documents list and pick your export method. You may save it as a PDF, email it, or upload it to the cloud.
pdfFiller makes dealing with documents a breeze. Create an account to find out!
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out describe form orbitals using

How to fill out and describe form orbitals using:
01
Understand the concept of orbitals: Before filling out a form and describing orbitals, it is important to have a basic understanding of what orbitals are. Orbitals are the regions of space around an atom where electrons are likely to be found. They have specific shapes and energy levels.
02
Gather necessary information: Before filling out the form, gather all the necessary information about the orbitals you want to describe. This may include the atomic number, electron configuration, and other relevant data.
03
Familiarize yourself with the form: Read and understand the form you are using to describe the orbitals. Pay attention to the specific sections and questions related to orbitals. This will ensure that you provide accurate and appropriate information.
04
Start with the basics: Begin filling out the form by providing the basic information about the atom for which you are describing the orbitals. This may include the atomic number, element symbol, and electron configuration.
05
Describe the shape of orbitals: In the form, there may be sections to describe the shape of the orbitals. Use appropriate notations, such as s, p, d, or f, to indicate the different types of orbitals. Provide details about their shapes, including the number of lobes, orientation, and any other relevant characteristics.
06
Provide energy level information: Many orbitals have different energy levels. Use the form to describe the energy levels of the orbitals you are discussing. This could involve providing information about the principal quantum number or specific energy values.
07
Include any additional information: If the form provides space for additional information or comments, use it to further describe the orbitals. You may include details about electron spin, orbital diagrams, or any other relevant information.
Who needs to describe form orbitals using:
01
Chemistry students: Students studying chemistry, especially at advanced levels, may need to fill out forms and describe orbitals as part of their coursework. Understanding orbitals and how to describe them is crucial for a thorough understanding of chemical bonding and reactivity.
02
Researchers and scientists: Professionals working in the field of chemistry, particularly in areas such as quantum mechanics or materials science, may need to describe orbitals on a regular basis. This information is often necessary for their research and experimental investigations.
03
Teachers and educators: Teachers who are teaching chemistry or related subjects may need to guide their students in filling out and describing form orbitals. They need to be familiar with the process themselves in order to effectively teach their students.
In conclusion, filling out and describing form orbitals requires a solid understanding of the concept of orbitals, gathering necessary information, and accurately providing details about the shape, energy level, and other characteristics of the orbitals. Individuals such as chemistry students, researchers, scientists, and educators may all benefit from this knowledge and need to describe form orbitals using.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
What is describe form orbitals using?
Describe form orbitals using is used to provide a detailed explanation of the orbital shapes and characteristics.
Who is required to file describe form orbitals using?
Science students and researchers who are studying atomic structure and electron arrangement are required to file describe form orbitals using.
How to fill out describe form orbitals using?
To fill out describe form orbitals using, one must carefully analyze the electron configuration data and use it to describe the spatial distribution of electrons in an atom.
What is the purpose of describe form orbitals using?
The purpose of describe form orbitals using is to enhance understanding of atomic structure and electron arrangement by visually representing the shapes and orientations of atomic orbitals.
What information must be reported on describe form orbitals using?
Information such as the number of electrons in each orbital, the shape of the orbitals, and the orientation of the orbitals must be reported on describe form orbitals using.
How can I modify describe form orbitals using without leaving Google Drive?
By integrating pdfFiller with Google Docs, you can streamline your document workflows and produce fillable forms that can be stored directly in Google Drive. Using the connection, you will be able to create, change, and eSign documents, including describe form orbitals using, all without having to leave Google Drive. Add pdfFiller's features to Google Drive and you'll be able to handle your documents more effectively from any device with an internet connection.
How can I get describe form orbitals using?
The premium pdfFiller subscription gives you access to over 25M fillable templates that you can download, fill out, print, and sign. The library has state-specific describe form orbitals using and other forms. Find the template you need and change it using powerful tools.
How do I edit describe form orbitals using on an iOS device?
Use the pdfFiller mobile app to create, edit, and share describe form orbitals using from your iOS device. Install it from the Apple Store in seconds. You can benefit from a free trial and choose a subscription that suits your needs.
Fill out your describe form orbitals using online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Describe Form Orbitals Using is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.