Last updated on Apr 6, 2026

Get the free Colony Stimulating Factors PreDetermination Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is colony stimulating factors predetermination
The Colony Stimulating Factors PreDetermination Form is a healthcare document used by physicians to request authorization for specific medications necessary for cancer treatment or neutropenia management.
pdfFiller scores top ratings on review platforms




Who needs colony stimulating factors predetermination?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to colony stimulating factors predetermination
What is the Colony Stimulating Factors PreDetermination Form?
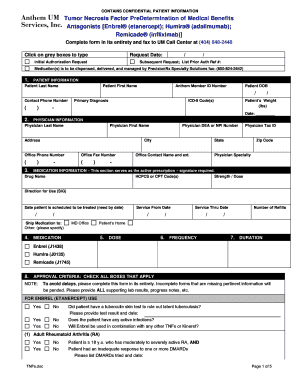
The Colony Stimulating Factors PreDetermination Form is an essential document in the healthcare sector, specifically designed for authorizing medications like Leukine®, Neulasta®, and Neupogen®. This form is particularly significant for patients undergoing cancer treatment or facing conditions that lead to neutropenia. By streamlining the authorization process, it aids healthcare providers in ensuring that patients receive necessary treatments in a timely manner.
This authorization form allows physicians to submit essential requests for treatment coverage, facilitating smoother interactions with insurance providers. Its importance is heightened for patients in critical medical situations that rely on colony stimulating factors.
Purpose and Benefits of the Colony Stimulating Factors PreDetermination Form
The primary purpose of the Colony Stimulating Factors PreDetermination Form is to provide a structured way for healthcare providers to ensure timely access to required medications. With this form, physicians can promptly request authorizations necessary for insurance coverage, thus minimizing delays in treatment onset.
Another benefit is adherence to local regulations in Georgia, ensuring that all necessary steps comply with applicable healthcare laws. This preparation allows healthcare providers to focus more on patient care rather than administrative hurdles.
Key Features of the Colony Stimulating Factors PreDetermination Form
The Colony Stimulating Factors PreDetermination Form features several critical elements that streamline its use. Required fields include patient demographic information, medication details, and clinical criteria that must be met for approval. Each section is designed to ensure that comprehensive and accurate information is provided.
Furthermore, the form mandates physician completion and signature, enhancing the validity of submissions. These requirements help maintain the integrity of the preauthorization process, ensuring that healthcare professionals meet necessary standards for medication requests.
Who Needs the Colony Stimulating Factors PreDetermination Form?
This form is particularly relevant for healthcare providers and physicians involved in the treatment of patients with specific medical conditions requiring colony stimulating factors. Those treating patients with cancer or complications of neutropenia will find this form integral to their practice.
Understanding eligibility for the form is crucial, as it is specifically tailored for individuals undergoing treatments involving CSFs. Physicians must be aware of these requirements to facilitate effective patient care and form usage.
How to Fill Out the Colony Stimulating Factors PreDetermination Form Online (Step-by-Step)
To efficiently complete the Colony Stimulating Factors PreDetermination Form online, follow these steps:
-
Access the form through pdfFiller.
-
Gather essential patient and physician information, including medication history and specific treatment details.
-
Fill in all required fields accurately, checking for completeness.
-
Ensure all clinical criteria are met and that the form is signed by the prescribing physician.
-
Review the filled form for errors before submission.
Review and Validation Checklist for the Colony Stimulating Factors PreDetermination Form
To facilitate correct form completion, consider the following checklist:
-
Verify that all mandatory fields are filled out.
-
Double-check that necessary supporting materials are attached.
-
Avoid common errors such as incomplete information or missing signatures.
-
Review the entire form to ensure accuracy before submission.
Submission Methods and Delivery
After completing the Colony Stimulating Factors PreDetermination Form, several methods are available for submission. Common options include faxing the form to the UM Call Center. It is important to adhere to submission timeframes to ensure prompt processing of the authorization request.
Be aware of any associated fees or payment methods that may be required during the submission process. Awareness of these details can help prevent delays in authorization and treatment access.
What Happens After You Submit the Colony Stimulating Factors PreDetermination Form
Once the Colony Stimulating Factors PreDetermination Form has been submitted, it undergoes a thorough review process. During this phase, the submission may be accepted, marked for further information, or rejected.
Healthcare providers are advised to track the status of their preauthorization requests vigilantly. Understanding common reasons for rejection can aid in timely corrections or amendments to submissions, enhancing chances of approval.
Ensure Your Security and Compliance When Using the Colony Stimulating Factors PreDetermination Form
When utilizing the Colony Stimulating Factors PreDetermination Form, implementing strong security measures is crucial. pdfFiller employs advanced security protocols, ensuring compliance with HIPAA and data protection standards during form completion.
Healthcare providers should take proactive steps to safeguard patient information when submitting forms. This responsibility enhances overall trust in the handling of sensitive documents.
Experience Seamless Form Management with pdfFiller
pdfFiller offers a user-friendly interface that facilitates the editing and eSigning of medical forms, including the Colony Stimulating Factors PreDetermination Form. This cloud-based platform allows users to fill out and manage documents conveniently and securely.
By utilizing pdfFiller, healthcare providers can benefit from comprehensive document management functionalities, enhancing efficiency in their practice while ensuring data compliance and security.
How to fill out the colony stimulating factors predetermination
-
1.To begin, access pdfFiller and locate the Colony Stimulating Factors PreDetermination Form in the search bar or by browsing the forms library.
-
2.Once you’ve found the form, open it in the pdfFiller editor, where you can easily navigate through the fields associated with patient and physician information.
-
3.Before starting the completion, gather all necessary details such as patient demographics, medication specifics, and any relevant clinical criteria that must be included in the form.
-
4.As you fill out the form, utilize the checkboxes provided for clinical criteria and ensure all mandatory fields are completed with accurate information.
-
5.After filling in the details, carefully review each section to confirm that the information entered is correct and complete, aiming to avoid common errors.
-
6.Once reviewed, arrange for the prescribing physician to sign the form electronically within the pdfFiller interface, ensuring it meets the requirement for authorization.
-
7.Finally, save your completed form by selecting the download option to keep a copy for your records, and submit it by faxing to the UM Call Center as per the instructions provided.

Who is eligible to use the Colony Stimulating Factors PreDetermination Form?
This form is intended for healthcare providers, particularly physicians who prescribe Colony Stimulating Factors, such as oncologists and other specialists treating patients with neutropenia.
What is the deadline for submitting the Colony Stimulating Factors PreDetermination Form?
While specific deadlines may vary depending on the insurance provider, it’s recommended to submit the form as early as possible to avoid delays in medication approval and treatment.
How do I submit the Colony Stimulating Factors PreDetermination Form?
The completed form must be faxed to the UM Call Center after the physician’s signature is obtained. Ensure compliance with any submission guidelines set by the provider.
What supporting documents are needed when submitting this form?
Typically, no additional documents are required with the Colony Stimulating Factors PreDetermination Form, but confirming with the healthcare provider's office or insurance company is advisable.
What common mistakes should I avoid when filling out this form?
Ensure that all fields are accurately completed, particularly physician and patient information. Remember to check required signatures and clinical criteria checks to prevent processing delays.
What are the processing times for approvals after submitting this form?
Processing times can vary; however, most insurance companies aim to provide a decision within a few business days. Check with the specific insurer for their timeline.
Is notarization required for the Colony Stimulating Factors PreDetermination Form?
No, notarization is not required for this form, but it must be signed by the prescribing physician before submission.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.