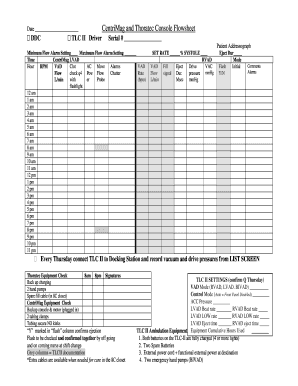
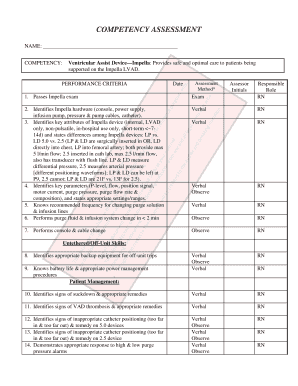
Get the free Roadmap for study startup
Show details
How-To Guide Roadmap for study startup Deploying Adobe technology to automate clinical study startup procedures Section 1: Introduction and overview 2 1.1 Introduction 2 1.2 Overview of the clinical
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign roadmap for study startup

Edit your roadmap for study startup form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.

Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your roadmap for study startup form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing roadmap for study startup online
In order to make advantage of the professional PDF editor, follow these steps below:
1
Log in to your account. Start Free Trial and register a profile if you don't have one yet.
2
Simply add a document. Select Add New from your Dashboard and import a file into the system by uploading it from your device or importing it via the cloud, online, or internal mail. Then click Begin editing.
3
Edit roadmap for study startup. Rearrange and rotate pages, add new and changed texts, add new objects, and use other useful tools. When you're done, click Done. You can use the Documents tab to merge, split, lock, or unlock your files.
4
Get your file. When you find your file in the docs list, click on its name and choose how you want to save it. To get the PDF, you can save it, send an email with it, or move it to the cloud.
With pdfFiller, it's always easy to deal with documents. Try it right now
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out roadmap for study startup

How to fill out a roadmap for study startup:
01
Identify the objectives: Clearly define the goals and objectives of the study startup. This will help in determining the necessary steps and milestones.
02
Determine the timeline: Set realistic timeframes for each stage of the study startup process. Consider factors like regulatory approvals, recruitment of participants, data collection, and analysis.
03
Outline the tasks: Break down the study startup process into individual tasks and activities. This includes obtaining ethics approval, recruiting and training study personnel, developing study protocols, and establishing data management systems.
04
Assign responsibilities: Clearly define who is responsible for each task or activity. Assign roles to relevant team members, such as principal investigators, research coordinators, and statisticians.
05
Establish checkpoints and milestones: Identify key checkpoints and milestones to track progress throughout the study startup. This helps ensure that the study is on track and allows for necessary adjustments if needed.
06
Determine resource requirements: Assess the resources needed to complete each task and allocate them accordingly. This includes personnel, funding, equipment, and materials.
07
Anticipate potential challenges: Identify potential challenges or roadblocks that may arise during the study startup process. Develop contingency plans to address these challenges and minimize their impact on the study.
08
Review and revise: Regularly review and update the roadmap as the study startup progresses. This allows for flexibility and adjustments based on new information or changes in circumstances.
Who needs a roadmap for study startup:
01
Researchers: Researchers who are initiating a new study or project will benefit from having a roadmap for study startup. It helps in planning and organizing the necessary steps and tasks.
02
Research coordinators: Research coordinators play a critical role in the study startup process. They are responsible for ensuring that all necessary approvals are obtained, protocols are developed, and study personnel are trained. A roadmap can help them stay organized and track progress.
03
Funding agencies: Funding agencies that provide grants or support for research studies may require a roadmap for study startup as part of the grant application or approval process. It allows them to assess the feasibility and soundness of the study plan.
04
Regulatory bodies: Regulatory bodies, such as ethics committees and institutional review boards, may request a roadmap for study startup to ensure that all necessary approvals and procedures are in place before the study begins. It helps in ensuring the protection of participants and adherence to ethical guidelines.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How can I edit roadmap for study startup from Google Drive?
Simplify your document workflows and create fillable forms right in Google Drive by integrating pdfFiller with Google Docs. The integration will allow you to create, modify, and eSign documents, including roadmap for study startup, without leaving Google Drive. Add pdfFiller’s functionalities to Google Drive and manage your paperwork more efficiently on any internet-connected device.
How can I edit roadmap for study startup on a smartphone?
The best way to make changes to documents on a mobile device is to use pdfFiller's apps for iOS and Android. You may get them from the Apple Store and Google Play. Learn more about the apps here. To start editing roadmap for study startup, you need to install and log in to the app.
How do I complete roadmap for study startup on an Android device?
Use the pdfFiller mobile app and complete your roadmap for study startup and other documents on your Android device. The app provides you with all essential document management features, such as editing content, eSigning, annotating, sharing files, etc. You will have access to your documents at any time, as long as there is an internet connection.
What is roadmap for study startup?
The roadmap for study startup is a strategic plan that outlines the key steps and milestones necessary to initiate a research study efficiently and effectively.
Who is required to file roadmap for study startup?
Typically, study sponsors, clinical research organizations, and principal investigators are required to file a roadmap for study startup to ensure compliance with regulatory requirements and organizational practices.
How to fill out roadmap for study startup?
To fill out the roadmap for study startup, follow outlined guidelines which include detailing timelines, identifying stakeholders, assigning responsibilities, and specifying key milestones and deliverables.
What is the purpose of roadmap for study startup?
The purpose of the roadmap for study startup is to provide a clear framework for planning, coordinating, and overseeing the startup phase of a study, ensuring all necessary steps are completed on time.
What information must be reported on roadmap for study startup?
The information reported on the roadmap for study startup usually includes study objectives, timelines, resource allocation, compliance requirements, and critical milestones that need to be achieved.
Fill out your roadmap for study startup online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Roadmap For Study Startup is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.