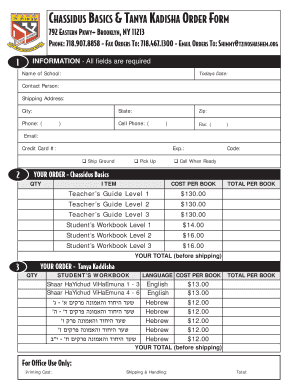
Get the free Product & Process / Systems Change Management Process
Show details
This document outlines the agenda, minutes, and action items related to the Systems Change Management Process (CMP) and various change requests for the Qwest Systems. It includes detailed discussions
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign product process systems change

Edit your product process systems change form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.

Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your product process systems change form via URL. You can also download, print, or export forms to your preferred cloud storage service.
How to edit product process systems change online
To use the services of a skilled PDF editor, follow these steps below:
1
Create an account. Begin by choosing Start Free Trial and, if you are a new user, establish a profile.
2
Prepare a file. Use the Add New button. Then upload your file to the system from your device, importing it from internal mail, the cloud, or by adding its URL.
3
Edit product process systems change. Add and change text, add new objects, move pages, add watermarks and page numbers, and more. Then click Done when you're done editing and go to the Documents tab to merge or split the file. If you want to lock or unlock the file, click the lock or unlock button.
4
Get your file. Select your file from the documents list and pick your export method. You may save it as a PDF, email it, or upload it to the cloud.
Dealing with documents is always simple with pdfFiller.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out product process systems change

How to fill out Product & Process / Systems Change Management Process
01
Gather necessary documentation related to the product or process.
02
Identify the key stakeholders involved in the change management process.
03
Define the scope and objectives of the change.
04
Assess the current state of the product or process that will be changed.
05
Develop a detailed change plan outlining the steps required for implementation.
06
Communicate the change plan to all stakeholders and gather their feedback.
07
Implement the change in phases, if possible, to minimize disruption.
08
Monitor the implementation closely to address any issues that arise.
09
Evaluate the success of the change post-implementation.
10
Document lessons learned and update relevant processes for future changes.
Who needs Product & Process / Systems Change Management Process?
01
Managers seeking to improve product efficiency.
02
Process improvement teams aiming to enhance operational workflows.
03
Stakeholders involved in implementing systematic organizational changes.
04
Quality assurance teams needing to ensure compliance with industry standards.
05
Employees affected by the changes for training and adaptation.
06
External partners and suppliers who may be impacted by the new processes.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
What is Product & Process / Systems Change Management Process?
The Product & Process / Systems Change Management Process is a structured approach to managing changes in products and processes within an organization. It involves assessing, approving, and implementing changes to ensure that they align with business objectives and meet regulatory requirements.
Who is required to file Product & Process / Systems Change Management Process?
Typically, employees or teams involved in product development, quality assurance, and compliance must file the Product & Process / Systems Change Management Process when proposing changes. This includes engineers, product managers, and quality control personnel.
How to fill out Product & Process / Systems Change Management Process?
To fill out the Product & Process / Systems Change Management Process, you should provide detailed information about the proposed change, including the nature of the change, reasons for the change, potential impacts on current operations, and any relevant documentation or analysis that supports the change.
What is the purpose of Product & Process / Systems Change Management Process?
The purpose of the Product & Process / Systems Change Management Process is to ensure that any changes to products or processes are systematically evaluated, approved, and documented to minimize risks and ensure compliance with industry standards and regulations.
What information must be reported on Product & Process / Systems Change Management Process?
The information that must be reported includes a description of the change, the rationale behind it, the affected products or processes, any potential impacts, risk assessments, implementation plans, and any necessary stakeholder approvals.
Fill out your product process systems change online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Product Process Systems Change is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.