
Get the free Sign off sheet for - California Department of Public Health - cdph ca
Show details
Status: California Department of Health Services Version: 2.0 SH/PKP/LCM Food and Drug Laboratory Branch (FDB) Microbiology Section Procedure PCR-1000 Page 1 of 9 TITLE: Detection of Shiva toxin-producing
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign sign off sheet for
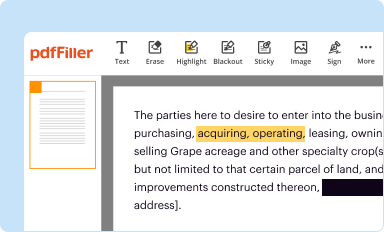
Edit your sign off sheet for form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
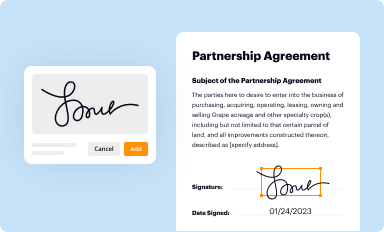
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your sign off sheet for form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing sign off sheet for online
Use the instructions below to start using our professional PDF editor:
1
Log in to account. Click on Start Free Trial and register a profile if you don't have one yet.
2
Upload a document. Select Add New on your Dashboard and transfer a file into the system in one of the following ways: by uploading it from your device or importing from the cloud, web, or internal mail. Then, click Start editing.
3
Edit sign off sheet for. Rearrange and rotate pages, insert new and alter existing texts, add new objects, and take advantage of other helpful tools. Click Done to apply changes and return to your Dashboard. Go to the Documents tab to access merging, splitting, locking, or unlocking functions.
4
Get your file. When you find your file in the docs list, click on its name and choose how you want to save it. To get the PDF, you can save it, send an email with it, or move it to the cloud.
pdfFiller makes working with documents easier than you could ever imagine. Register for an account and see for yourself!
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out sign off sheet for
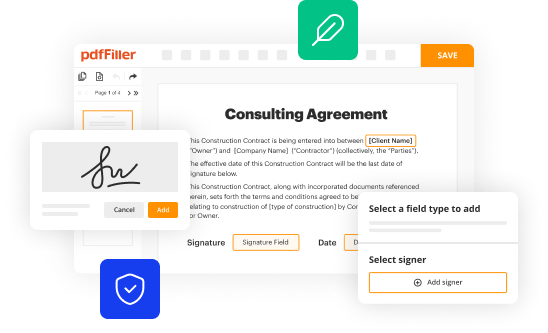
To fill out a sign off sheet, follow these steps:
01
Write the date and time of the sign off.
02
Clearly state the project or task that is being signed off.
03
Provide a brief description of the completed work or deliverable.
04
Include any necessary notes or comments regarding the work or any issues encountered.
05
Sign and print your name to indicate your approval or acknowledgment.
A sign off sheet is typically needed by:
01
Project Managers: They use sign off sheets to document the completion of project milestones or deliverables.
02
Team Leaders: They require sign off sheets to track the progress of their team members and ensure tasks are completed.
03
Contractors or Vendors: They may need sign off sheets as evidence of completing their contractual obligations.
Remember, the use of sign off sheets may vary depending on the industry and specific project requirements.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How do I edit sign off sheet for online?
pdfFiller allows you to edit not only the content of your files, but also the quantity and sequence of the pages. Upload your sign off sheet for to the editor and make adjustments in a matter of seconds. Text in PDFs may be blacked out, typed in, and erased using the editor. You may also include photos, sticky notes, and text boxes, among other things.
How can I fill out sign off sheet for on an iOS device?
Install the pdfFiller app on your iOS device to fill out papers. If you have a subscription to the service, create an account or log in to an existing one. After completing the registration process, upload your sign off sheet for. You may now use pdfFiller's advanced features, such as adding fillable fields and eSigning documents, and accessing them from any device, wherever you are.
How do I complete sign off sheet for on an Android device?
Use the pdfFiller mobile app to complete your sign off sheet for on an Android device. The application makes it possible to perform all needed document management manipulations, like adding, editing, and removing text, signing, annotating, and more. All you need is your smartphone and an internet connection.
What is sign off sheet for?
A sign off sheet is a document used to collect acknowledgement or confirmation from individuals involved in a particular task or project, indicating their approval or agreement.
Who is required to file sign off sheet for?
The individuals who are directly involved in a task or project are usually required to file the sign off sheet.
How to fill out sign off sheet for?
To fill out a sign off sheet, individuals need to provide their name, role, date, and signature, indicating their approval or agreement.
What is the purpose of sign off sheet for?
The purpose of a sign off sheet is to ensure that all necessary parties have reviewed and approved a task or project, minimizing potential misunderstandings or disputes.
What information must be reported on sign off sheet for?
The sign off sheet typically requires the following information: name of the individual, their role, date, and signature.
Fill out your sign off sheet for online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Sign Off Sheet For is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.





















