Last updated on Apr 12, 2026

Get the free Preliminary Screening Part II Form 202
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is preliminary screening part ii
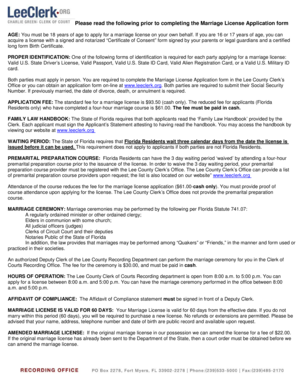
The Preliminary Screening Part II Form 202 is a medical history form used by certified research staff to collect health condition information and medical treatments for incontinence to determine eligibility for study participation.
pdfFiller scores top ratings on review platforms




Who needs preliminary screening part ii?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to preliminary screening part ii
What is the Preliminary Screening Part II Form 202?
The Preliminary Screening Part II Form 202 is a crucial document in healthcare, primarily focused on collecting data related to incontinence eligibility for research participation. This form aids certified UITN research staff in evaluating potential participants' qualifications for studies. It ensures that all necessary health information is systematically gathered, enhancing the integrity of the research process.
By utilizing the preliminary screening form, researchers can obtain vital data that helps in understanding the health conditions affecting patients, which contributes to informed decision-making in medical studies.
Purpose and Benefits of the Preliminary Screening Part II Form 202
This form plays an essential role for both researchers and patients. It streamlines the collection of medical history and health condition data, which is imperative for assessing a patient's eligibility to participate in incontinence research studies. By employing a standardized approach, researchers benefit from consistent data that enhances the quality and reliability of outcomes.
Patients also reap advantages from this standardized data collection as it clarifies their medical conditions and available treatment options, ultimately guiding their healthcare decisions.
Key Features of the Preliminary Screening Part II Form 202
-
Contains sections for personal information and comprehensive medical history.
-
Includes fillable fields like 'Study ID Number' and 'Date Interview Completed'.
-
Requires the recording of 'Consent obtained' and other critical details.
-
Features checkboxes for quick assessment of eligibility criteria.
The patient screening form is designed to facilitate efficient data entry, ensuring that all relevant medical information is readily available for review.
Who Needs the Preliminary Screening Part II Form 202?
The primary audience for the Preliminary Screening Part II Form 202 includes researchers and healthcare professionals involved in incontinence studies. It is also intended for patients who meet the eligibility criteria for these research studies.
Accurate and thorough data collection is vital for achieving effective health outcomes, making this form an indispensable tool for professionals in the healthcare field.
How to Fill Out the Preliminary Screening Part II Form 202 Online
Filling out the form online using pdfFiller is straightforward. Follow these steps:
-
Access the Preliminary Screening Part II Form 202 via pdfFiller.
-
Enter the 'Study ID Number' and 'Date Interview Completed'.
-
Complete all fillable sections, ensuring accuracy in your responses.
-
Review the form to confirm that all required fields are completed.
-
Submit the form electronically through the platform.
This ease of use and accessibility makes pdfFiller an excellent choice for filling out medical treatment forms.
Common Errors and How to Avoid Them When Using the Form
When completing the Preliminary Screening Part II Form 202, individuals often make several common mistakes. These include leaving required fields blank or providing unclear information. To enhance clarity and accuracy, consider the following tips:
-
Double-check each section of the form before submission.
-
Ensure that all required fields are filled out completely and accurately.
Taking the time to verify information will minimize errors and ensure that data is correctly represented.
Security and Compliance Considerations for the Preliminary Screening Part II Form 202
Data security is paramount when managing sensitive medical information. The Preliminary Screening Part II Form 202 adheres to strict compliance standards, including HIPAA and GDPR. pdfFiller employs robust security features to protect user data, assuring individuals that their information remains confidential throughout the submission process.
Utilizing a platform that prioritizes security reinforces trust and compliance when handling personal medical documents.
How to Submit the Preliminary Screening Part II Form 202
There are several methods available for submitting the Preliminary Screening Part II Form 202, including:
-
Online submission via pdfFiller.
-
Direct mail to the designated research facility (if applicable).
After submission, users can expect a confirmation and, where necessary, guidance on tracking the status of their form.
Real-Life Example of a Completed Preliminary Screening Part II Form 202
To assist users in visualizing the process, a real-life example of a completed form is beneficial. Each section should be filled out meticulously, demonstrating how information is collected effectively and accurately.
This guided example highlights the importance of adhering to form requirements, ensuring that prospective participants understand what is expected during data entry.
Experience the Ease of Filling Out the Preliminary Screening Part II Form 202 with pdfFiller
Using pdfFiller for the Preliminary Screening Part II Form 202 offers numerous advantages, such as eSigning capabilities and document management features. The platform's user-friendly interface allows individuals to edit and share forms efficiently.
Starting with pdfFiller ensures a seamless and secure experience in form-filling processes, promoting effective management of medical documentation while maintaining privacy and compliance.
How to fill out the preliminary screening part ii
-
1.Access pdfFiller and log into your account.
-
2.Search for 'Preliminary Screening Part II Form 202' in the document library.
-
3.Once you find the form, click to open it in the editor.
-
4.Familiarize yourself with the template structure, noting the fillable fields and sections.
-
5.Before you begin filling the form, gather required documents such as previous medical history records and any notes from patient interviews.
-
6.Start by entering the 'Study ID Number' and 'Date Interview Completed' in the designated fields.
-
7.For the 'Interviewer's Initials' section, input the initials of the authorized interviewer.
-
8.In the section marked 'Consent obtained', confirm consent has been secured by checking the appropriate box.
-
9.Proceed to fill out sections regarding general information, eligibility, health conditions, and treatments based on collected data.
-
10.If additional information is required, ensure you consult with relevant medical records or interview notes.
-
11.Review all entries for accuracy, ensuring all required fields are filled completely.
-
12.Once you have completed all sections, utilize the pdfFiller functions to finalize the document.
-
13.Save the form locally or select to download it in your preferred format once completed.
-
14.If required, submit the form electronically through pdfFiller or follow up with your institution's submission process.

What are the eligibility requirements for this form?
Eligibility typically involves having a relevant medical history related to incontinence. Only patients who meet specific criteria for the study can participate, and consent must be obtained before filling out this form.
Is there a deadline for submitting the form?
Submission deadlines may vary based on the study protocol. Always check with your research coordinator for specific timelines to ensure timely submission of the form.
How should I submit the completed form?
The completed form can be submitted via pdfFiller's electronic submission process, or you may need to print and submit it according to your organization's guidelines. Confirm submission methods with your study coordinator.
What supporting documents are required?
You may need to include medical records, interview notes, and consent documents to support the information provided in the form. Ensure all relevant details are collected before starting.
What are common mistakes to avoid when filling this form?
Common mistakes include incomplete fields, incorrect data entry, and failing to secure proper consent. Double-check each section for accuracy and completeness before submission.
How long does processing take once submitted?
Processing times can vary, but typically it takes a few days to a week for review. Contact your research team for specifics related to your study.
Can I edit the form after it is completed?
Yes, if you are using pdfFiller, you can edit the form after completion until you finalize and submit it. Always double-check for any necessary changes before you submit.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.