Last updated on Apr 10, 2026

Palmetto GBA Form M00116 free printable template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Palmetto GBA Form M00116
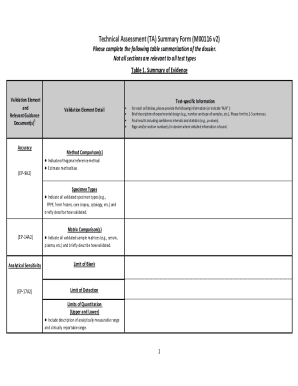
The Technical Assessment Summary Form M00116 is a medical records release document used by healthcare professionals to summarize key evidence and validation elements in technical assessments.
pdfFiller scores top ratings on review platforms




Who needs Palmetto GBA Form M00116?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Palmetto GBA Form M00116
What is the Technical Assessment Summary Form M00116?
The Technical Assessment Summary Form M00116 serves a critical function in healthcare assessments, summarizing evidence and validation elements essential for technical evaluations. This form is applicable to a variety of healthcare technical assessments, providing a structured approach to documentation. Its primary purpose lies in consolidating vital information regarding experimental design, results, and relevant validation criteria.
Purpose and Benefits of the Technical Assessment Summary Form M00116
Utilizing the Technical Assessment Summary Form M00116 in healthcare and research facilitates improved organization of data, thereby enhancing the validation process. This medical validation form helps ensure compliance with established standards, leading to more efficient workflows. Additionally, using pdfFiller for filling and managing this healthcare validation form enables users to streamline operations and maintain high accuracy in documentation.
Key Features of the Technical Assessment Summary Form M00116
The form includes distinct fields that cater to various sections, such as experimental design, results, and analysis. Key functionality for users encompasses:
-
Ease of editing and eSigning via pdfFiller.
-
Special formatting options, like tables, to organize data effectively.
These features reinforce the form's role as a technical assessment template and enhance overall usability.
Who Needs the Technical Assessment Summary Form M00116?
This form is primarily targeted towards medical professionals, researchers, and institutional bodies involved in healthcare evaluations. Specific scenarios where the healthcare form M00116 is necessary include validation studies and compliance checks with healthcare standards. It's crucial for users to understand its importance in meeting legal and regulatory requirements, particularly for clinical validation purposes.
How to Fill Out the Technical Assessment Summary Form M00116 Online
To fill out the Technical Assessment Summary Form M00116 online using pdfFiller, follow these steps:
-
Access the form through the pdfFiller platform.
-
Complete the required fields, ensuring accuracy.
-
Pay attention to validation hints provided for each section.
Common mistakes to avoid include skipping mandatory fields and providing unclear information that may impede the assessment process.
Submission Methods and Delivery
Users can submit the completed Technical Assessment Summary Form M00116 through various channels. Options include online submissions directly through pdfFiller and email delivery. Compliance with specific protocols may be necessary, depending on the healthcare setting. Typical timelines for processing submissions should also be taken into consideration to ensure adherence to operational schedules.
Security and Compliance When Using the Technical Assessment Summary Form M00116
Handling sensitive documents securely is paramount, especially in healthcare settings. pdfFiller adheres to stringent security measures, ensuring compliance with HIPAA and GDPR regulations while employing robust data encryption practices. Additionally, users should be aware of record retention requirements and privacy standards that govern the management of technical assessment documents.
Sample of a Completed Technical Assessment Summary Form M00116
A visual example or detailed description of a filled-out Technical Assessment Summary Form M00116 can enhance user comprehension. Highlighting best practices for accurately completing each section will guide users in utilizing the template effectively. Resources available on pdfFiller can further assist users in understanding how to utilize a medical validation form correctly.
Successfully Managing Your Technical Assessment Summary Form M00116
After submission, users should actively manage their Technical Assessment Summary Form M00116 by tracking its status and preparing for potential follow-ups. Key practices include:
-
Maintaining copies for personal records.
-
Understanding common reasons for rejection to reduce future issues.
These steps will help ensure that the process remains efficient and organized.
Achieving Efficiency with pdfFiller’s Technical Assessment Summary Form M00116
By utilizing pdfFiller, users can achieve a seamless form-filling experience, benefiting from features such as online editing, eSigning, and comprehensive form tracking. This integration significantly enhances the efficiency of various form management tasks, catering specifically to the needs of healthcare professionals managing the Technical Assessment Summary Form M00116.
How to fill out the Palmetto GBA Form M00116
-
1.Access the Technical Assessment Summary Form M00116 on pdfFiller by clicking on the provided link.
-
2.Open the form, and familiarize yourself with the layout, which includes fields and checkboxes designed for your responses.
-
3.Before completing the form, gather all necessary information related to the technical assessment, including details on experimental design and outcomes.
-
4.Begin filling in the required fields, ensuring that you answer all questions accurately based on your data.
-
5.Utilize pdfFiller's features to navigate between sections easily, and checkboxes for validation elements.
-
6.Once you have completed the form, take the time to review your entries for any inaccuracies or missing data.
-
7.Use the preview feature to see how the final document will appear once finalized.
-
8.After reviewing, save your work by clicking the save button, and download the completed form in your preferred format.
-
9.If necessary, submit the form through the provided submission options available on pdfFiller.

Who is eligible to use the Technical Assessment Summary Form M00116?
The Technical Assessment Summary Form M00116 is primarily designed for healthcare professionals, researchers, and administrators involved in medical validation studies.
What is the deadline for submitting the Technical Assessment Summary Form?
Submission deadlines vary by study or organizational requirements. It is best to check with your supervising authority or organization for specific timelines.
How can I submit the completed form once filled out?
You can submit the completed Technical Assessment Summary Form M00116 through your institutional channels, or if using pdfFiller, leverage the submission options available in the platform.
What supporting documents are required to complete the form?
Typically, documentation related to your technical assessment, including experimental results and validation data, should accompany the completed form.
What are common mistakes to avoid when filling out the form?
Common mistakes include overlooking required fields, providing incomplete information, and failing to follow the form's instructions for summarizing data accurately.
How long does it take to process the form after submission?
Processing times can vary based on the organization or authority reviewing the technical assessment. Allow sufficient time for feedback or updates.
Is notarization required for the Technical Assessment Summary Form?
No, notarization is not required for the Technical Assessment Summary Form M00116, making it easier to complete and submit.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.