Last updated on Apr 10, 2026

Immunization Action Coalition P3041 free printable template
pdfFiller is not affiliated with any government organization
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Immunization Action Coalition P3041
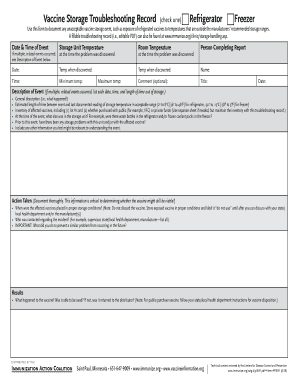
The Vaccine Storage Troubleshooting Record is a healthcare document used by professionals to document any unacceptable vaccine storage events and ensure compliance with storage guidelines.
pdfFiller scores top ratings on review platforms




Who needs Immunization Action Coalition P3041?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Immunization Action Coalition P3041
What is the Vaccine Storage Troubleshooting Record?
The Vaccine Storage Troubleshooting Record is a critical form utilized in healthcare settings to document any unacceptable vaccine storage events, such as exposure to improper temperatures. This record plays a pivotal role in ensuring compliance with storage guidelines and maintaining vaccine safety.
It serves the dual purpose of documenting vaccine storage events and promoting accountability among healthcare professionals. Adhering to these guidelines is essential for ensuring both safety and compliance, especially in environments where vaccine efficacy is paramount.
-
Definition of the Vaccine Storage Troubleshooting Record.
-
Explanation of its purpose in documenting vaccine storage events.
-
Importance of adhering to vaccine storage guidelines for safety and compliance.
Purpose and Benefits of Documenting Vaccine Storage Events
Documenting vaccine storage events is crucial for healthcare professionals. Keeping a detailed record directly impacts patient safety and ensures the efficacy of vaccines. This practice not only protects patients but also aids organizations in complying with necessary healthcare regulations.
Comprehensive documentation acts as a safety net, providing insights into any incidents that could compromise vaccine integrity. By maintaining accurate records, healthcare providers can swiftly address and rectify storage issues, thereby upholding the highest standards of care.
-
Benefits of maintaining records of vaccine storage events.
-
Impact on patient safety and vaccine efficacy.
-
Aid in compliance with healthcare regulations.
Key Features of the Vaccine Storage Troubleshooting Record
The Vaccine Storage Troubleshooting Record boasts several essential features designed for ease of use in healthcare environments. Critical fields included in the record feature event details, affected vaccines, and actions taken to remedy the situation.
Designed for monitoring vaccine temperatures, this document is crucial for ensuring that vaccines remain in the appropriate storage conditions. Moreover, the record's digital accessibility through platforms like pdfFiller enhances usability and efficiency.
-
List of essential fields included in the record.
-
Explanation of how to utilize it for monitoring vaccine temperatures.
-
Mention of digital accessibility and ease of use.
Who Needs the Vaccine Storage Troubleshooting Record?
This important form is primarily utilized by healthcare professionals, including nurses and pharmacists, who are responsible for documenting storage events. Understanding eligibility criteria for using the form is vital for compliance and quality assurance within healthcare settings.
Moreover, compliance officers and quality assurance teams benefit from this form, as it aids their roles in overseeing that storage protocols are correctly implemented and followed across facilities.
-
Overview of healthcare professionals required to use the form.
-
Eligibility criteria for use within healthcare settings.
-
Importance of the form for compliance officers and quality assurance teams.
How to Fill Out the Vaccine Storage Troubleshooting Record Online
Completing the Vaccine Storage Troubleshooting Record requires attention to detail. Detailed instructions on each field are provided to guide users in entering necessary data accurately. Before filling out the form, it is advisable to compile pertinent information to minimize errors.
A checklist of required information can help streamline the completion process. Additionally, users should be aware of common errors to avoid to ensure the form is filled out correctly.
-
Step-by-step guide on completing the form.
-
Checklist of information needed to gather beforehand.
-
Common errors to avoid.
Submission Process for the Vaccine Storage Troubleshooting Record
The submission process for the Vaccine Storage Troubleshooting Record is designed to be straightforward. Multiple digital submission methods are available through pdfFiller, which simplifies the process for users. Tracking submissions and confirmation processes provide added peace of mind during this step.
Additionally, users must be aware of any potential fees or deadlines associated with their submissions to ensure compliance with organizational requirements.
-
Overview of digital submission methods available.
-
Information on tracking submissions and confirmations.
-
Discussion of potential fees and deadlines.
Security and Compliance when Handling Vaccine Storage Records
When handling sensitive documents like the Vaccine Storage Troubleshooting Record, security and compliance are paramount. pdfFiller ensures all data protection measures align with HIPAA compliance and industry standards. Secure document management protects patient information and promotes trust in healthcare processes.
Utilizing pdfFiller facilitates safe record-keeping, ensuring that sensitive information remains confidential throughout its lifecycle.
-
Overview of data protection measures and HIPAA compliance.
-
Importance of secure document management in healthcare.
-
How pdfFiller ensures safety during record-keeping.
What Happens After You Submit the Vaccine Storage Troubleshooting Record
After you submit the Vaccine Storage Troubleshooting Record, it is essential to remain informed about the next steps. Users should expect a certain processing time and be ready for any feedback during the review phase. Understanding how to correct or amend a submitted record is important should any issues arise.
Common reasons for rejection may also exist, and knowing how to address these concerns can enhance the overall documentation process.
-
Insights into processing time and what to expect during review.
-
How to correct or amend a submitted record if necessary.
-
Common reasons for rejection and how to address them.
Common Scenarios and Examples of Completed Vaccine Storage Troubleshooting Records
Providing practical examples helps users better understand the Vaccine Storage Troubleshooting Record. Sample scenarios that illustrate potential vaccine storage issues are invaluable for training and reference. Additionally, including examples of completed records can inform best practices for maintaining accurate documentation.
Maintaining precise records is crucial for compliance and quality assurance in vaccine management protocols.
-
Sample scenarios illustrating potential vaccine storage issues.
-
Examples of filled-out records for reference.
-
Discussion of best practices for maintaining accurate records.
Experience Seamless Document Management with pdfFiller
pdfFiller offers a robust platform for users to efficiently complete the Vaccine Storage Troubleshooting Record while ensuring compliance and security. The features available for editing, filling, eSigning, and submitting the form enhance user experience.
Its user-friendly design allows for easy accessibility from any browser, making the document management journey both practical and secure for all users in need of effective solutions.
-
Highlight pdfFiller's features for document management.
-
Emphasize user-friendly design and accessibility.
-
Encourage users to leverage pdfFiller for secure and efficient documentation.
How to fill out the Immunization Action Coalition P3041
-
1.Access pdfFiller and search for the Vaccine Storage Troubleshooting Record form in the template library.
-
2.Once located, click on the form to open it within the pdfFiller interface.
-
3.Gather necessary information prior to filling out the form, including details of the storage event, affected vaccines, and actions taken.
-
4.Navigate through the form using the fillable fields. Click on each field to enter data, ensuring all relevant details are documented.
-
5.Provide detailed descriptions for each unacceptable storage event, including temperature readings, affected vaccines, and any immediate actions taken.
-
6.Once all fields are completed, review the information to ensure there are no errors or omissions.
-
7.Use pdfFiller’s tools to check the completeness of the form, ensuring all required sections are filled in correctly.
-
8.After finalizing the form, save your work within pdfFiller, choosing an appropriate file name for easy reference.
-
9.If needed, download the completed form as a PDF for printing or submit it electronically if your organization requires it.

Who is eligible to use the Vaccine Storage Troubleshooting Record?
The Vaccine Storage Troubleshooting Record can be utilized by healthcare professionals, including vaccine coordinators, pharmacists, and public health officials, involved in vaccine storage management.
When should I complete the Vaccine Storage Troubleshooting Record?
You should complete the form immediately after any unacceptable vaccine storage event occurs, such as when vaccines are exposed to incorrect temperatures, to ensure accurate documentation.
How do I submit the Vaccine Storage Troubleshooting Record?
Submission methods may vary by organization. Generally, you can download the completed record and submit it via email, or upload it to your organization's preferred storage system.
What supporting documents are required with this form?
Typically, no additional documents are required when submitting the Vaccine Storage Troubleshooting Record. However, including any relevant temperature logs or incident reports can enhance clarity.
What are common mistakes to avoid when filling out this form?
Ensure all fields are filled completely, double-check the accuracy of temperature readings and descriptions, and avoid leaving blank spaces unless stated otherwise.
How long is the Vaccine Storage Troubleshooting Record kept?
The retention period for the record may differ by organization, but it is advisable to retain it for at least three years to comply with regulatory guidelines.
What should I do if I make an error on the form?
If you make an error on the Vaccine Storage Troubleshooting Record, simply correct the mistake in the fillable field. Always review the completed form before finalizing to prevent errors.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.