Last updated on Mar 28, 2026

Get the free IV Injectable Drug Review Request Form
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is iv injectable drug review
The IV Injectable Drug Review Request Form is a healthcare document used by patients and providers to request a review for IV therapy or injectable drugs.
pdfFiller scores top ratings on review platforms




Who needs iv injectable drug review?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to iv injectable drug review
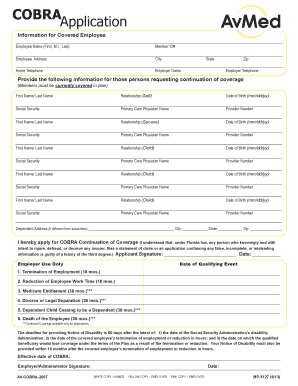
What is the IV Injectable Drug Review Request Form?
The IV Injectable Drug Review Request Form serves a vital role in healthcare by facilitating the approval of intravenous (IV) therapy or injectable drugs. This form is crucial for requesting authorization from HMSA in Hawaii, allowing healthcare providers to ensure that patients receive necessary medications promptly. It is specifically designed to streamline the approval process, making it easier for providers to access needed treatments for their patients.
Purpose and Benefits of the IV Injectable Drug Review Request Form
Utilizing the IV Injectable Drug Review Request Form offers several key advantages in the healthcare landscape. It aids in accelerating the drug approval process, ultimately enhancing patient care by ensuring timely access to treatments. Furthermore, this form fosters effective communication between healthcare providers and insurance companies, minimizing misunderstandings and expediting the authorization process.
-
Streamlined drug approval for patient care
-
Enhanced communication between healthcare providers and insurers
Who Needs the IV Injectable Drug Review Request Form?
This form is essential for various stakeholders in the healthcare system, including healthcare providers, patients, and insurers. Specific situations that warrant the use of this form include cases where prior authorization is required for injectable drugs or IV therapy. Understanding who needs the form assists in ensuring that all necessary parties can facilitate the approval process efficiently.
-
Healthcare providers seeking drug approval for patients
-
Patients in need of designated treatments
-
Insurance companies needing documentation for coverage
Eligibility Criteria for Submitting the IV Injectable Drug Review Request Form
Eligibility to submit the IV Injectable Drug Review Request Form is determined by specific criteria, including membership requirements. Individuals or entities eligible to submit this form typically include active members of HMSA. Certain conditions or diagnoses might also necessitate the use of this form, ensuring that it is utilized appropriately and effectively.
-
Active HMSA membership required
-
Specific diagnoses that warrant IV therapy
How to Fill Out the IV Injectable Drug Review Request Form Online (Step-by-Step)
Follow these steps to fill out the IV Injectable Drug Review Request Form online accurately:
-
Enter the membership number in the designated field.
-
Fill in the patient’s name and pertinent details.
-
Provide the diagnosis using the ICD-9/ICD-10 code.
-
List the medication name and dosage required.
-
Attach any necessary supporting documentation as instructed.
Key Features of the IV Injectable Drug Review Request Form
The IV Injectable Drug Review Request Form includes several critical elements necessary for effective submission. Key sections feature space for diagnosis, provider information, and documentation requirements. To enhance accuracy and completeness, providers should pay close attention to all entries and ensure all relevant information is included before submission.
-
Sections for diagnosis and treatment information
-
Provider contact details
-
Requirements for supporting documentation
Submission Process for the IV Injectable Drug Review Request Form
Submitting the IV Injectable Drug Review Request Form involves several straightforward steps. Once the form is completed, it should be faxed to the Medical Management Department of HMSA. Providers should be aware of submission timelines to avoid delays and understand what to expect post-submission regarding approval notifications.
-
Complete the form accurately
-
Fax the form to the designated number
-
Monitor for approval notifications
Common Errors and How to Avoid Them
Errors in completing the IV Injectable Drug Review Request Form can cause significant delays. Common mistakes include missing information and incorrect coding. To avoid these, healthcare providers should double-check their entries and ensure all required documentation is attached to the form before submission.
-
Verify all patient and drug information
-
Ensure that correct diagnosis codes are used
Security and Compliance Considerations for Handling the IV Injectable Drug Review Request Form
When handling the IV Injectable Drug Review Request Form, the importance of security cannot be overstated. Sensitive medical information must be submitted securely to protect patient privacy. pdfFiller complies with HIPAA and data protection standards, ensuring that all submissions are handled securely and responsibly.
Experience Simplified with pdfFiller for Your IV Injectable Drug Review Request Form
pdfFiller is an ideal solution for filling out, eSigning, and submitting the IV Injectable Drug Review Request Form with ease. Its user-friendly tools offer a secure way to manage healthcare forms, providing peace of mind in the submission process. Leveraging pdfFiller can enhance workflow efficiency while maintaining security for sensitive documents.
How to fill out the iv injectable drug review
-
1.Access pdfFiller and search for the 'IV Injectable Drug Review Request Form' to begin.
-
2.Open the form within pdfFiller's editor interface for easy navigation.
-
3.Ensure you have all necessary information handy including your Membership Number, Patient's Name, ICD-9/ICD-10 Code, and Drug Name before starting.
-
4.Fill in the blank fields with accurate details, ensuring that all required sections are completed.
-
5.If specific sections require supporting documentation, prepare these documents for attachment.
-
6.Review your entries carefully for accuracy, checking each completed section of the form.
-
7.Finalize the form by following pdfFiller's instructions to submit or print, ensuring no fields are left blank.
-
8.Save your completed form by clicking the download button or submit it to the appropriate party as instructed.

Who is eligible to use the IV Injectable Drug Review Request Form?
Patients and their healthcare providers in Hawaii seeking approval for IV therapies or injectable drugs are eligible to use this form.
What supporting documents are required for submission?
You'll need to attach relevant medical records or documents that demonstrate the necessity of the requested treatment. Ensure these are ready before starting the form.
How do I submit the completed form?
Once you have filled out the form on pdfFiller, you can submit it directly through the platform or print it out for faxing to HMSA's Medical Management Department.
What common mistakes should I avoid when completing this form?
Ensure all required fields are filled, double-check the accuracy of the ICD codes, and confirm that supporting documentation is attached before submission.
What is the processing time for the requests made using this form?
Processing times can vary. Typically, it may take a few business days, so it is advisable to submit the form as early as possible to avoid delays in treatment.
Is notarization required for the IV Injectable Drug Review Request Form?
No, notarization is not required for this form. Simply complete and submit it through the provided channels.
What should I do if I have questions about the form completion?
If you have questions, refer to the guidelines provided on pdfFiller or contact HMSA's Medical Management Department for further assistance.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.