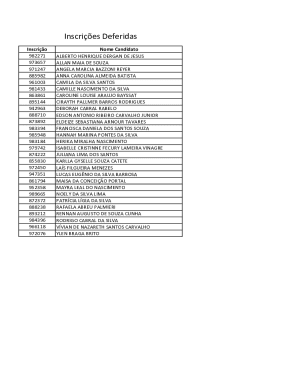
Get the free CIRB Clinical Trial Protocol Template - ClinicalTrials.gov
Show details
CLINICAL TRIAL PROTOCOL TITLE:
A pragmatic randomized controlled trial of a novel TCM physician involved collaborative care
model in the management of patients with axial Spondyloarthritis in Singapore
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign cirb clinical trial protocol

Edit your cirb clinical trial protocol form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.

Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your cirb clinical trial protocol form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing cirb clinical trial protocol online
Follow the steps down below to benefit from a competent PDF editor:
1
Create an account. Begin by choosing Start Free Trial and, if you are a new user, establish a profile.
2
Simply add a document. Select Add New from your Dashboard and import a file into the system by uploading it from your device or importing it via the cloud, online, or internal mail. Then click Begin editing.
3
Edit cirb clinical trial protocol. Add and replace text, insert new objects, rearrange pages, add watermarks and page numbers, and more. Click Done when you are finished editing and go to the Documents tab to merge, split, lock or unlock the file.
4
Save your file. Select it from your records list. Then, click the right toolbar and select one of the various exporting options: save in numerous formats, download as PDF, email, or cloud.
With pdfFiller, it's always easy to work with documents.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out cirb clinical trial protocol

How to fill out cirb clinical trial protocol
01
To fill out a CIRB clinical trial protocol, follow these steps:
02
Begin by providing the title of the protocol.
03
Include the identification number or code for the trial.
04
Write a brief introduction and background information about the trial.
05
State the objectives and aims of the trial.
06
Describe the study design and methodology.
07
Include the eligibility criteria for participants.
08
Specify the intervention or treatment being tested.
09
Outline the procedures and assessments to be conducted.
10
Provide details about the randomization and blinding, if applicable.
11
Explain the data collection and analysis plan.
12
Describe the potential risks and benefits of participation.
13
Include information about patient confidentiality and data protection.
14
Write a detailed plan for monitoring and quality assurance.
15
Specify the anticipated timeline for the trial.
16
Include any additional documents or appendices as necessary.
17
Review the completed protocol for accuracy and completeness.
18
Submit the filled-out CIRB clinical trial protocol for review and approval by the relevant authorities.
19
Make any revisions or updates as required.
20
Once approved, proceed with conducting the clinical trial according to the protocol.
21
Ensure ongoing adherence to the protocol and regular reporting of progress and results.
Who needs cirb clinical trial protocol?
01
CIRB clinical trial protocol is needed by researchers and sponsors who wish to conduct clinical trials involving human participants.
02
It is also required by regulatory authorities and ethics committees to ensure that the trial is conducted ethically, with participant safety and well-being as the top priority.
03
Medical professionals involved in the trial, such as investigators and study coordinators, also need the protocol as a guide for conducting the study.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
Where do I find cirb clinical trial protocol?
It's simple with pdfFiller, a full online document management tool. Access our huge online form collection (over 25M fillable forms are accessible) and find the cirb clinical trial protocol in seconds. Open it immediately and begin modifying it with powerful editing options.
How do I execute cirb clinical trial protocol online?
Completing and signing cirb clinical trial protocol online is easy with pdfFiller. It enables you to edit original PDF content, highlight, blackout, erase and type text anywhere on a page, legally eSign your form, and much more. Create your free account and manage professional documents on the web.
Can I edit cirb clinical trial protocol on an Android device?
Yes, you can. With the pdfFiller mobile app for Android, you can edit, sign, and share cirb clinical trial protocol on your mobile device from any location; only an internet connection is needed. Get the app and start to streamline your document workflow from anywhere.
What is cirb clinical trial protocol?
The CIRB clinical trial protocol is a detailed plan that outlines the objectives, design, methodology, statistical considerations, and organization of a clinical trial.
Who is required to file cirb clinical trial protocol?
Researchers conducting clinical trials are required to file the CIRB clinical trial protocol.
How to fill out cirb clinical trial protocol?
The CIRB clinical trial protocol should be filled out by providing all the necessary information as outlined in the protocol template provided by the institution or regulatory body.
What is the purpose of cirb clinical trial protocol?
The purpose of the CIRB clinical trial protocol is to ensure that the trial is conducted safely, ethically, and in accordance with regulatory requirements.
What information must be reported on cirb clinical trial protocol?
The CIRB clinical trial protocol must include details on the study design, patient population, intervention, endpoints, statistical analysis, and ethical considerations.
Fill out your cirb clinical trial protocol online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Cirb Clinical Trial Protocol is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.