Last updated on Dec 24, 2014
Get the free Laboratory Service Request Bioassay Testing
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Bioassay Testing Request
The Laboratory Service Request Bioassay Testing is a healthcare form used by clients to request bioassay testing services from Pacific BioLabs.
pdfFiller scores top ratings on review platforms




Who needs Bioassay Testing Request?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Bioassay Testing Request
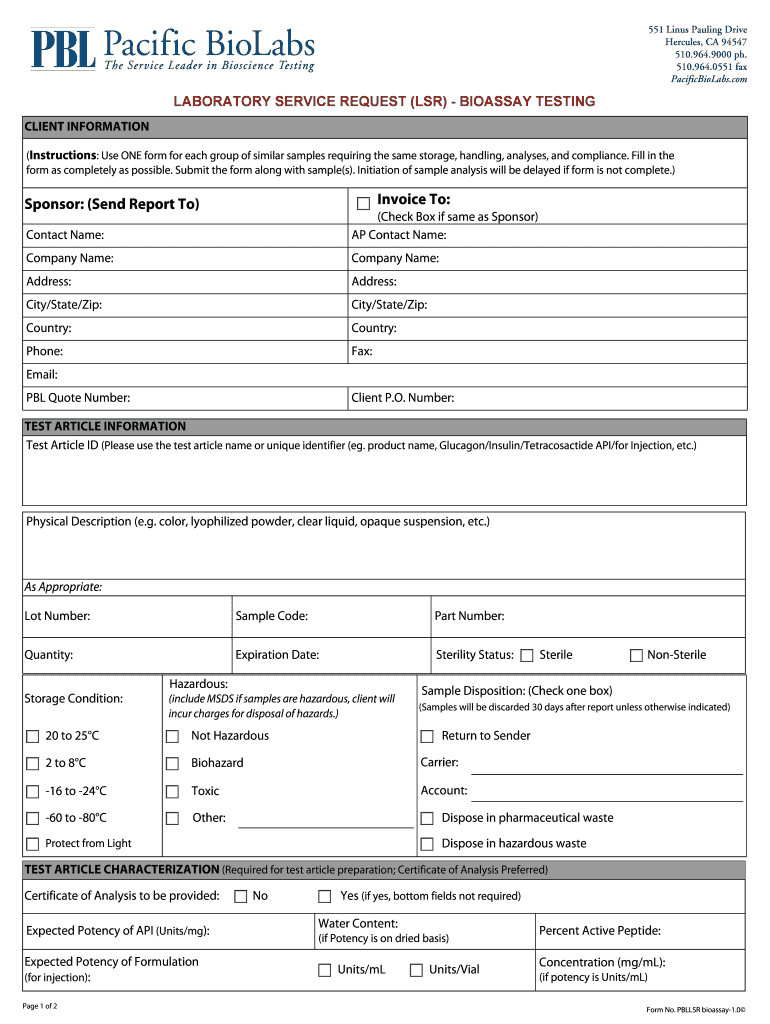
What is the Laboratory Service Request Bioassay Testing?
The Laboratory Service Request (LSR) for bioassay testing serves as a formal document clients use to detail specific testing requirements at Pacific BioLabs. Its primary purpose is to streamline the process of requesting various bioassay tests critical for research and product development. Compliance with regulations plays a vital role, as the sponsor is responsible for ensuring that the submission adheres to both internal and external policies.
By completing the laboratory service request form accurately, clients facilitate the testing procedure and maintain adherence to industry standards. This not only enhances the reliability of results but also ensures that bioassay testing aligns with the necessary regulatory guidelines.
Purpose and Benefits of the Laboratory Service Request Bioassay Testing
The Laboratory Service Request is essential for bioassay testing, providing a systematic approach for clients to document their needs. The benefits of utilizing this request are numerous, including improved accuracy and reliability of test results, which are crucial for regulatory compliance.
Clients may find this testing form especially invaluable during critical phases of research or product development, such as clinical trials or quality control assessments. Scenarios may include validating the efficacy of a new drug or ensuring the safety of a biopharmaceutical product, where adherence to regulated standards is crucial.
Key Features of the Laboratory Service Request Bioassay Testing
Understanding the key features of the LSR bioassay testing form is crucial for effective utilization. The form includes several fillable fields, such as Contact Name, Company Name, and Test Article ID, which are essential for submission accuracy. Additionally, it outlines testing purposes, specifies storage conditions, and clarifies compliance requirements necessary for successful processing.
Another pivotal feature is the signature line, which serves to authenticate the request and ensure accountability. This signature indicates that the sponsor has reviewed the details and authorizes the bioassay testing.
Who Needs the Laboratory Service Request Bioassay Testing?
Potential users of the Laboratory Service Request include research sponsors and laboratories engaged in bioassay testing. Various industries, such as pharmaceuticals, biotechnology, and agricultural research, require bioassay testing to comply with regulatory demands and validate product safety and efficacy.
Eligibility criteria for using the Laboratory Service Request typically include having a valid reason for testing, such as research and development objectives. This form is vital for any entity looking to ensure that their testing is conducted in accordance with established protocols.
How to Fill Out the Laboratory Service Request Bioassay Testing Online (Step-by-Step)
Filling out the Laboratory Service Request bioassay testing form online can be done effectively using pdfFiller. Here are the steps to complete the form:
-
Access the form using pdfFiller.
-
Input your Contact Name and Company Name accurately.
-
Provide the required information in the Test Article section, including its ID and description.
-
Specify storage conditions and any additional instructions as needed.
-
Ensure the form is signed by the authorized sponsor before submission.
Best practices include verifying all entries for accuracy and completeness to avoid unnecessary delays in processing.
Common Errors and How to Avoid Them When Submitting the Laboratory Service Request Bioassay Testing
Many common errors arise during the completion of the Laboratory Service Request form, which can impede processing. Frequent mistakes include incomplete fields, incorrect contact information, and failure to include a required signature.
To minimize errors, clients should review the form thoroughly before submission. Double-checking compliance requirements and confirming that the sponsor's authorization is included are essential steps to ensure the request moves smoothly through the review process.
Submission Methods and Delivery for the Laboratory Service Request Bioassay Testing
Clients have several options for submitting the Laboratory Service Request. Most notably, electronic submission through pdfFiller provides a convenient and efficient way to send requests. Additionally, physical copies can also be submitted through traditional channels if necessary.
Clients should consider tracking submissions and obtaining confirmation of receipt to ensure their request has been properly processed and will receive the appropriate attention.
What Happens After You Submit the Laboratory Service Request Bioassay Testing?
After submitting the Laboratory Service Request, clients can expect a standard processing timeline, during which their request will be reviewed by the Pacific BioLabs team. Confirmation of receipt will be provided, outlining the next steps in the bioassay testing process.
Follow-up actions may include clarifications needed for the request or additional information if there are questions regarding the testing requirements. Maintaining open communication with Pacific BioLabs post-submission is crucial for ensuring timely processing.
Security and Compliance for the Laboratory Service Request Bioassay Testing
Security measures in place while using pdfFiller are designed to protect sensitive data associated with the Laboratory Service Request. Strict adherence to HIPAA and GDPR compliance is maintained throughout the handling of biomedical documents.
Ensuring data protection safeguards client confidentiality, which is integral in the processing of health-related forms, including medical records release forms.
Streamline Your Laboratory Service Request Bioassay Testing with pdfFiller
Utilizing pdfFiller for your Laboratory Service Request can greatly enhance efficiency. The platform offers user-friendly features such as editing capabilities, eSigning, and secure sharing options that simplify the entire process. Compared to traditional methods, pdfFiller streamlines the completion and management of the request form, making it convenient for all clients.
How to fill out the Bioassay Testing Request
-
1.Access the Laboratory Service Request Bioassay Testing form on pdfFiller's website by searching for the form name or using a direct link from your source.
-
2.Once the form is open, familiarize yourself with the interface. You will find fillable fields, checkboxes for options, and a signature line.
-
3.Before starting, gather the necessary information such as test article ID, physical description, lot number, and storage conditions, ensuring all details are accurate.
-
4.Begin filling out the form by clicking on the designated fields. Enter the contact name, company name, and address accurately in their respective sections.
-
5.Complete the sections related to the test article, elaborating on its characteristics and specific compliance requirements.
-
6.Make sure to check the boxes regarding testing purposes, regulatory compliance, and archive options as they apply to your request.
-
7.Once you have filled in all sections, thoroughly review the form for any errors or missing information.
-
8.Pay special attention to the signature line, where the sponsor must sign to authorize the testing. Ensure the correct representative is signing.
-
9.After finalizing the information, click on the save or submit option in pdfFiller to either download your completed form or submit it directly.
-
10.Choose to save the document in your desired format for your records, or share it with relevant stakeholders as needed.

Who is eligible to use the Laboratory Service Request Bioassay Testing form?
The form is designed for clients, such as biotech companies and pharmaceutical sponsors, who require bioassay testing services from Pacific BioLabs. Anyone authorized to manage these requests can complete the form.
Are there any specific deadlines for submitting this form?
While the form itself does not specify deadlines, it’s advisable to submit it as soon as possible to ensure timely testing and compliance with study timelines. Check with Pacific BioLabs for additional guidelines.
How should I submit the completed Laboratory Service Request form?
You can submit the form directly through pdfFiller, where you can electronically submit it to Pacific BioLabs, or download it and send via email or regular mail, as per your preference.
What supporting documents do I need to include with this form?
Typically, you may not need additional documents, but it's a good practice to include any relevant information related to the test article or regulatory requirements, depending on your specific request.
What are common mistakes to avoid when filling out this form?
Common mistakes include missing required fields, incorrect information about the test article, and not having the designated sponsor sign the form. Always double-check for completeness before submitting.
How long does it take to process the Laboratory Service Request form?
Processing times can vary based on the complexity of the testing required. Generally, after submission, you can expect feedback from Pacific BioLabs within a few business days.
Are there any fees associated with the bioassay testing request?
Fees for bioassay testing services are typically outlined by Pacific BioLabs. It is recommended to consult their pricing guidelines or contact their office directly for precise fee structures.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.