Last updated on Mar 24, 2026
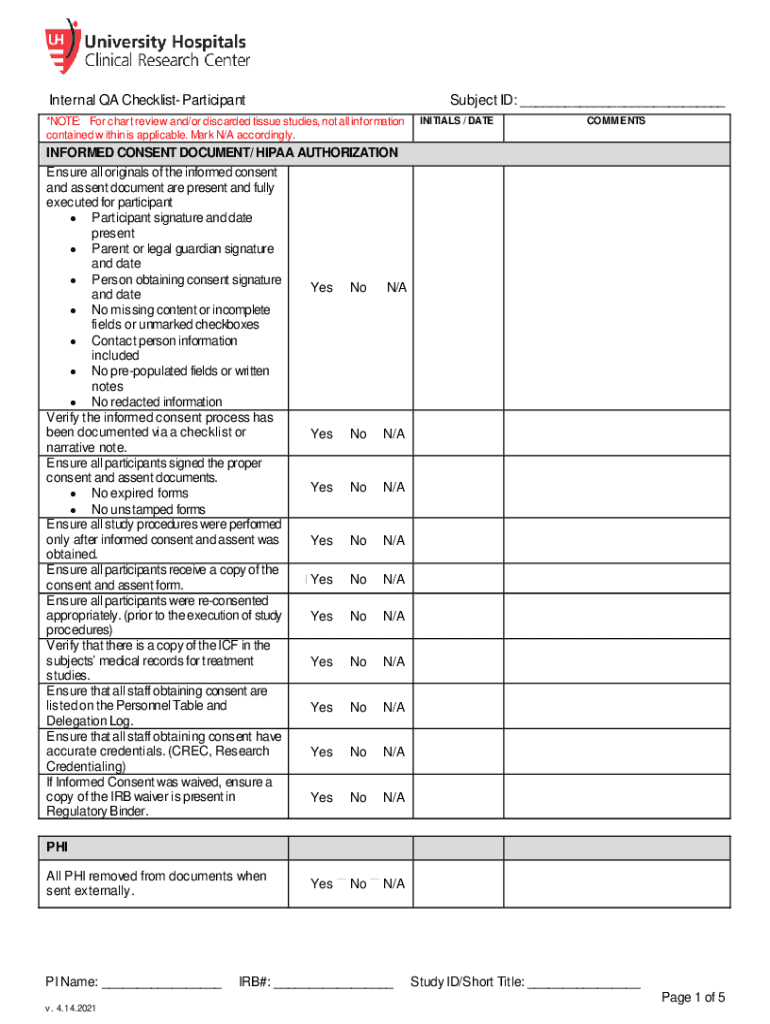
University Hospitals Internal QA Checklist free printable template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is University Hospitals Internal QA Checklist
The Internal QA Checklist Participant is a healthcare form used by participants in clinical studies to ensure all necessary consent and documentation processes are properly followed.
pdfFiller scores top ratings on review platforms




Who needs University Hospitals Internal QA Checklist?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to University Hospitals Internal QA Checklist
Understanding the Internal QA Checklist Participant: Purpose and Importance
The Internal QA Checklist Participant serves an essential role in healthcare consent processes. This document ensures that all stakeholders are informed and compliant with the requirements for clinical studies. Key to this process is the informed consent, which helps protect participants’ rights and welfare.
This checklist defines the roles involved in the signing process, ensuring each participant, parent, or legal guardian, and the person obtaining consent is accounted for. Active participation by all parties is crucial for maintaining integrity in clinical study documentation. This checklist is vital for upholding the standards of patient care in healthcare settings.
Key Features of the Internal QA Checklist Participant
The Internal QA Checklist Participant comprises various sections designed to streamline the consent process. It includes components such as informed consent details, eligibility verification, and safety reporting.
-
Informed Consent: Provides comprehensive details that participants need to understand.
-
Eligibility Verification: Confirms participant qualifications for the clinical study.
-
Safety Reporting: Ensures proper documentation of health and safety measures.
The form also features fillable fields, checkboxes, and signature requirements, making it user-friendly and efficient for clinical study checklist completion.
Who Needs to Complete the Internal QA Checklist Participant?
Several users must complete the Internal QA Checklist Participant, primarily participants themselves, parents or legal guardians, and the individual obtaining consent. Each role is essential to ensure that all necessary consents are correctly documented and validated.
This checklist must be utilized in various situations where informed consent is required, ensuring that each user fulfills their responsibilities accurately. Accurate completion by all parties helps prevent delays in clinical study processes and minimizes the risk of issues arising from incomplete or incorrect documentation.
How to Complete the Internal QA Checklist Participant Form Online
Completing the Internal QA Checklist Participant form online is a straightforward process. Follow these steps to ensure a smooth experience using pdfFiller:
-
Gather necessary information regarding the participant and study details.
-
Navigate to the online form using pdfFiller's user-friendly interface.
-
Fill in the required fields, ensuring all information is accurate.
-
Utilize the digital signing feature to authenticate the form.
By following these guidelines, users can effectively manage their data collection form process and facilitate safety reporting requirements.
Common Errors and How to Avoid Them When Filling Out the Internal QA Checklist
While filling out the Internal QA Checklist Participant form, several common mistakes can occur. Missing signatures, incomplete fields, and incorrect information are frequent pitfalls that can lead to delays in processing.
To avoid these issues, it is essential to review the form thoroughly before submission. Here are some tips to ensure accuracy:
-
Double-check all signatures and dates for completeness.
-
Confirm that all required fields are filled out appropriately.
A careful review can prevent common rejection reasons during the consent collection process.
Security and Compliance Considerations for the Internal QA Checklist Participant
When handling sensitive healthcare documents, security and compliance are critical. pdfFiller employs robust encryption methods and adheres to HIPAA and GDPR regulations, ensuring that all user data is adequately protected.
Data protection is especially important for healthcare forms, where confidentiality and security are paramount. Users can rest assured that their information is safeguarded during the entire process of filling out and submitting the checklist.
The Process After Submitting the Internal QA Checklist Participant
After submitting the Internal QA Checklist Participant, users can expect a confirmation of submission. Tracking the status of the checklist is vital for ensuring that clinical study processes proceed smoothly.
If any corrections or amendments are required after submission, users should be aware of the options available to address these changes quickly. Maintaining accurate records post-submission is crucial for all stakeholders involved in the clinical study.
Utilizing pdfFiller for Your Internal QA Checklist Participant Needs
pdfFiller offers a range of advantages for users completing the Internal QA Checklist Participant. Its features simplify the form-filling process, allowing for easy editing, electronic signing, and efficient cloud-based document management.
By using pdfFiller, users can take advantage of a seamless experience and begin filling out their healthcare consent form today. The platform's commitment to user-friendly tools enhances the overall efficiency of clinical study documentation.
How to fill out the University Hospitals Internal QA Checklist
-
1.To access the Internal QA Checklist Participant form on pdfFiller, visit their website and use the search bar to find the form by its name or category.
-
2.Once the form is open, navigate through the document using the toolbar on the left side of the screen. You can zoom in or out for better visibility.
-
3.Before filling out the form, gather all necessary information such as participant details, signatures, dates, and any relevant documentation that may be required.
-
4.Start completing the form by clicking on the designated fields. Enter the necessary participant information, ensuring to check all boxes that apply, such as eligibility verification and safety reporting.
-
5.After filling in all the required sections, take a moment to review each field for accuracy. Make sure that all signatures, including those of parents or guardians, are included.
-
6.Once the form is complete and all information is confirmed, you can finalize it. Use the 'Finish' button to go to the next step.
-
7.To save the completed form, choose the download option to save it as a PDF to your device or submit it directly through pdfFiller if instructed.

What are the eligibility requirements for using this form?
To use the Internal QA Checklist Participant form, individuals must be participants in a clinical study, parents or legal guardians of minors needing consent, or authorized personnel obtaining consent. Ensure all required parties are willing and eligible.
Are there deadlines for submitting this form?
Yes, it is crucial to complete and submit the Internal QA Checklist Participant form prior to any study procedures. Check with your study coordinator for specific deadlines to ensure compliance.
How can I submit the completed Internal QA Checklist Participant form?
The completed form can be submitted through pdfFiller via its submission options or directly to your study coordinator, depending on the instructions provided for your clinical study.
What supporting documents are required with this form?
Generally, no additional documents are required when submitting the Internal QA Checklist Participant form. However, ensure that all required signatures and participant information are accurately filled to avoid any issues.
What common mistakes should I avoid when filling out this form?
Common mistakes include leaving fields blank, missing signatures from necessary parties, and not verifying the accuracy of participant information. Review the form thoroughly before submission.
How long does processing of this form usually take?
Processing times for the Internal QA Checklist Participant form can vary based on the study's administrative procedures. It's advisable to confirm with your study coordinator for specific timelines.
Is there a fee associated with using this form?
Typically, there are no fees for using the Internal QA Checklist Participant form itself. However, you may want to check your specific clinical study's guidelines regarding any potential costs.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.



















