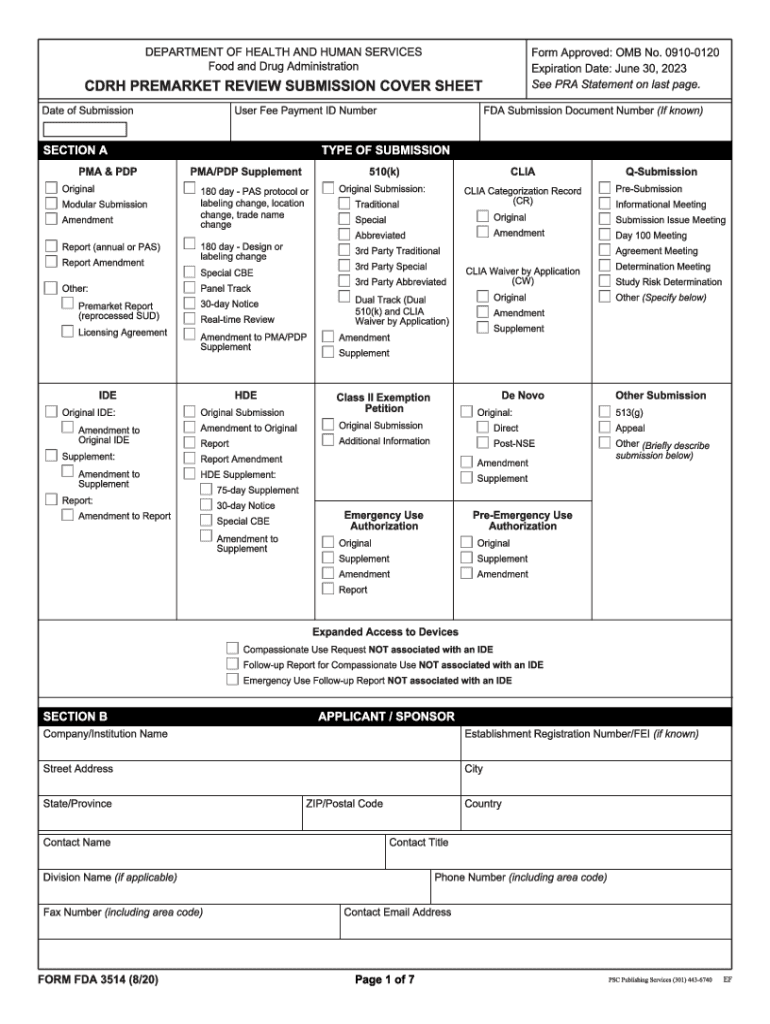
FDA 3514 2020-2026 free printable template
Show details
An agency may not conduct or sponsor, and a person is not required to respond to, a collection of information unless it displays a currently valid OMB control number.
pdfFiller is not affiliated with any government organization
Get, Create, Make and Sign pdffiller form
Edit your form 3514 fda form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your form fda 3514 form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing fda 3514 form online
Here are the steps you need to follow to get started with our professional PDF editor:
1
Set up an account. If you are a new user, click Start Free Trial and establish a profile.
2
Upload a document. Select Add New on your Dashboard and transfer a file into the system in one of the following ways: by uploading it from your device or importing from the cloud, web, or internal mail. Then, click Start editing.
3
Edit fda 3514 form. Rearrange and rotate pages, add and edit text, and use additional tools. To save changes and return to your Dashboard, click Done. The Documents tab allows you to merge, divide, lock, or unlock files.
4
Save your file. Select it from your list of records. Then, move your cursor to the right toolbar and choose one of the exporting options. You can save it in multiple formats, download it as a PDF, send it by email, or store it in the cloud, among other things.
It's easier to work with documents with pdfFiller than you can have believed. You may try it out for yourself by signing up for an account.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
FDA 3514 Form Versions
Version
Form Popularity
Fillable & printabley
How to fill out cdrh premarket review submission cover sheet form
How to fill out FDA 3514
01
Obtain FDA Form 3514 from the FDA website or another official source.
02
Begin filling out the form by providing your name, company name, and contact information in the designated fields.
03
Indicate the type of product you are submitting information for, such as a biological product or drug.
04
Complete the section that specifies the purpose of the submission, including the relevant regulations.
05
Provide a detailed description of the product, including its intended use and manufacturing process.
06
Include any necessary supporting documentation, such as clinical study data or manufacturing protocols.
07
Review the form for accuracy and completeness before submitting.
08
Submit the form as directed, either electronically or by mail, depending on FDA requirements.
Who needs FDA 3514?
01
Individuals or companies developing biological products, vaccines, or drugs that need to submit product information to the FDA.
02
Regulatory affairs professionals responsible for ensuring compliance with FDA regulations.
03
Researchers or manufacturers looking to gain approval or communicate information about their products to the FDA.
Fill
3514
: Try Risk Free






People Also Ask about how to fill out fda 3514 their products to the fda
Is FDA form 3514 required?
FDA Form 3514. The use of this form is optional. If you choose not to use the form, ensure that the relevant information is contained in the cover letter:.
What is FDA form 3514?
Form 3514) The CDRH Premarket Review Submission Cover Sheet11 is a voluntary form used to help provide basic administrative information for all types of premarket notification submissions.
What are the requirements for 510k reporting?
A 510(k) requires demonstration of substantial equivalence to another legally U.S. marketed device. Substantial equivalence means that the new device is as safe and effective as the predicate. the information submitted to FDA demonstrates that the device is as safe and effective as the legally marketed device.
What is FDA Form 3514?
Form 3514) The CDRH Premarket Review Submission Cover Sheet11 is a voluntary form used to help provide basic administrative information for all types of premarket notification submissions.
What is form 3454?
Form FDA 3454, or the Financial Certification or Disclosure Statement, is used to submit information regarding clinical investigators who participated in the clinical studies. If no clinical studies were performed, simply state: “no clinical studies were performed to test this device.”
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How do I make changes in fda form 3514 cdrh premarket review submission cover sheet?
With pdfFiller, the editing process is straightforward. Open your form fda 3514 cdrh premarket review submission cover sheet in the editor, which is highly intuitive and easy to use. There, you’ll be able to blackout, redact, type, and erase text, add images, draw arrows and lines, place sticky notes and text boxes, and much more.
Can I create an eSignature for the form 3514 in Gmail?
It's easy to make your eSignature with pdfFiller, and then you can sign your form 3514 business code must be entered right from your Gmail inbox with the help of pdfFiller's add-on for Gmail. This is a very important point: You must sign up for an account so that you can save your signatures and signed documents.
How do I edit fda form 3674 straight from my smartphone?
The pdfFiller mobile applications for iOS and Android are the easiest way to edit documents on the go. You may get them from the Apple Store and Google Play. More info about the applications here. Install and log in to edit fda template.
What is FDA 3514?
FDA 3514 is a form used by manufacturers of biologics to report product defects, failures, or adverse events associated with the use of biological products.
Who is required to file FDA 3514?
Manufacturers, importers, and healthcare providers who have knowledge of adverse events related to biological products are required to file FDA 3514.
How to fill out FDA 3514?
To fill out FDA 3514, you need to provide specific information regarding the product, the event occurrence, details of the reporting entity, and any relevant follow-up information.
What is the purpose of FDA 3514?
The purpose of FDA 3514 is to facilitate the reporting of adverse events and ensure the safety and effectiveness of biological products by collecting data for regulatory oversight.
What information must be reported on FDA 3514?
The information that must be reported on FDA 3514 includes the identity of the product, the nature of the adverse event, details about the patient, and any testing or evaluation performed.
Fill out your FDA 3514 online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Fda Form 3514 Download is not the form you're looking for?Search for another form here.
Keywords relevant to 3514 form
Related to form 3514 california
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.
























