Last updated on Mar 24, 2026

Canada PHSA Laboratories Normal Quality Control free printable template
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Canada PHSA Laboratories Normal Quality Control
The Urine Dipstick Form B1 - Quality Control Record Sheet is a healthcare document used by laboratories to record urine quality control test results.
pdfFiller scores top ratings on review platforms




Who needs Canada PHSA Laboratories Normal Quality Control?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Canada PHSA Laboratories Normal Quality Control
What is the Urine Dipstick Form B1 - Quality Control Record Sheet?
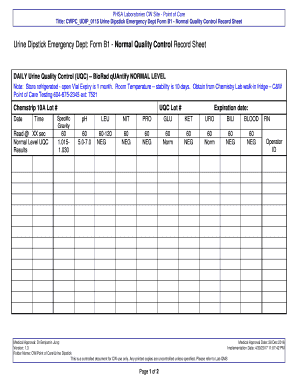
The Urine Dipstick Form B1 serves as an essential tool in medical laboratories, particularly for urine quality control testing. This form is specifically designed for use with the BioRad qUAntify® equipment, which allows for accurate testing and verification of urine samples. At PHSA Laboratories, it plays a crucial role in point of care testing, ensuring that results are reliable and consistent.
Used regularly in healthcare settings, this quality control record sheet helps standardize procedures, enhancing the overall effectiveness of monitoring urine quality. With its focus on accuracy, the Urine Dipstick Form B1 is vital in maintaining high standards in laboratory work.
Purpose and Benefits of the Urine Dipstick Form B1
Utilizing the Urine Dipstick Form B1 is beneficial for healthcare professionals and laboratories. Accurate urine quality control is pivotal for patient safety, enabling effective treatment decisions by ensuring that test results reflect true physiological conditions. Furthermore, standardized record-keeping supports compliance with regulations and improves operational efficiency.
This form also facilitates tracking test results over time, making it easier to identify potential issues in urine quality. The combination of these advantages contributes to improved patient outcomes and streamlined laboratory operations.
Key Features of the Urine Dipstick Form B1
The Urine Dipstick Form B1 includes several essential fields necessary for thorough documentation. Key components of the form encompass:
-
Date performed
-
Performed by
-
Lot numbers
In addition, various test result checkboxes are provided for parameters such as specific gravity, pH, LEU, NIT, PRO, GLU, KET, URO, BILI, and BLOOD. This structured design aids in comprehensive data entry, ensuring that all relevant information is captured accurately.
Who Needs the Urine Dipstick Form B1?
The primary users of the Urine Dipstick Form B1 include laboratory technicians, healthcare providers, and quality control personnel within healthcare facilities. Its relevance extends to compliance officers and laboratory managers, who rely on this form for maintaining quality assurance within their institutions.
Institutions involved in point of care testing also find the form essential, as it helps ensure that testing protocols are followed and results are recorded accurately, contributing to better patient care.
How to Fill Out the Urine Dipstick Form B1 Online (Step-by-Step)
Filling out the Urine Dipstick Form B1 online requires careful attention to detail. Follow these steps for accurate completion:
-
Access the online platform designated for the form.
-
Enter the date performed in the appropriate field.
-
Fill in the name of the person who performed the test.
-
Input the lot number and expiration date.
-
Check the boxes for specific test results including specific gravity, pH, and other parameters.
-
Review the completed information for accuracy before submission.
To enhance the process, consider using specific software that can streamline the completion of forms and provide user-friendly validation tips.
Review and Validation Checklist for the Urine Dipstick Form B1
Before finalizing the Urine Dipstick Form B1, it's crucial to perform a thorough review. Ensure that the following aspects are checked for completeness and accuracy:
-
All fields are filled out correctly, including dates and signatures.
-
Lot numbers correspond to the test kits used.
-
Verify the accuracy of test result entries.
Common errors can be mitigated through colleague verification or dual checks, particularly in larger laboratory settings, to promote accuracy and reliability in data collection.
Security and Compliance for the Urine Dipstick Form B1
When handling sensitive documentation such as the Urine Dipstick Form B1, data protection and compliance are paramount. pdfFiller implements robust security measures, including 256-bit encryption, to safeguard all user information.
Furthermore, adherence to HIPAA and GDPR guidelines ensures that sensitive patient data remains confidential. Best practices in maintaining data privacy are essential to uphold the integrity of test results and patient information.
How to Submit and Where to File the Urine Dipstick Form B1
Submitting the Urine Dipstick Form B1 can be done through various methods. Users should be aware of the differences between paper and electronic submissions:
-
Paper submissions may require mailing to specific departments within PHSA Laboratories.
-
Electronic submissions are often more efficient, providing immediate confirmation of receipt.
It's also important to note any deadlines for submission, ensuring compliance with laboratory protocols and timely data processing.
What Happens After You Submit the Urine Dipstick Form B1?
Once the Urine Dipstick Form B1 is submitted, users can expect certain processing timelines based on laboratory workflows. In the event that corrections or amendments are necessary, understanding the procedures for adjustments is crucial.
Implementing tracking measures ensures that submitted forms can be monitored, providing peace of mind for users awaiting confirmation and results.
Filling Out the Urine Dipstick Form B1 with pdfFiller
Using pdfFiller significantly enhances the process of filling out and managing the Urine Dipstick Form B1. With features like editing, eSigning, and easy sharing, users can effectively manage their documentation.
Accessibility is a key advantage of pdfFiller, allowing users to complete forms from any device without needing downloads. Moreover, pdfFiller’s commitment to document security ensures compliance with necessary regulations throughout the entire process.
How to fill out the Canada PHSA Laboratories Normal Quality Control
-
1.Access pdfFiller and log in to your account. Search for 'Urine Dipstick Form B1' in the template library to find the correct form.
-
2.Open the form by clicking on its title. Familiarize yourself with the layout and available fields.
-
3.Before filling out the form, gather all necessary data, including test dates, the person performing the test, and relevant lot numbers and expiration dates.
-
4.Begin filling in the 'Date Performed' field with the correct date of the quality control test.
-
5.In the 'Performed by' field, enter the name of the person completing the test.
-
6.Locate the 'Chemstrip10A Lot #' field and input the specific lot number associated with your testing materials.
-
7.Next, fill in the 'Expiration Date' field with the appropriate expiration date for the chemstrip.
-
8.Proceed to check off results in fields relating to specific gravity, pH, LEU, NIT, PRO, GLU, KET, URO, BILI, and BLOOD, marking each box as applicable according to your results.
-
9.Once all fields are completed, review the form carefully to ensure all entries are accurate and complete.
-
10.After confirmation, you may save your form as a pdf using the download option or submit directly through pdfFiller if required.

Who is eligible to use the Urine Dipstick Form B1?
The Urine Dipstick Form B1 is primarily intended for use by laboratory technicians and healthcare providers involved in quality control testing of urine samples.
What information is needed to complete the form?
You will need the date of the test, the individual's name performing the test, lot numbers, expiration dates, and test results for specific gravity, pH, and other parameters.
How can I submit the completed form?
You can submit the completed Urine Dipstick Form B1 by saving it as a PDF through pdfFiller and then emailing it to the relevant authority or printing it for physical submission as required.
Are there specific deadlines for submitting this form?
While there are typically no specific deadlines related to the Urine Dipstick Form B1, it is advised to complete and submit the form promptly after performing quality control testing.
What common mistakes should I avoid when filling out this form?
Common mistakes include overlooking required fields, incorrect entries in the lot number or expiration date, and failing to mark all applicable test results. Double-check your work to avoid errors.
How is the quality control data processed after submission?
After submission, the quality control data from the Urine Dipstick Form B1 is reviewed by quality assurance personnel to ensure compliance and accuracy in laboratory testing protocols.
Is notarization required for this form?
No, notarization is not required for the Urine Dipstick Form B1. It is a controlled document strictly for use within the pilot clinical workflow.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.