
HHS FDA 3632 2014 free printable template
Show details
Form Approved: OMB No. 0910-0025 Expiration Date: January 31, 2017, FORM FDA 3632 (2/14) Guide for Preparing Product Reports for Lasers and Products Containing Lasers Public reporting burden for this
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign HHS FDA 3632
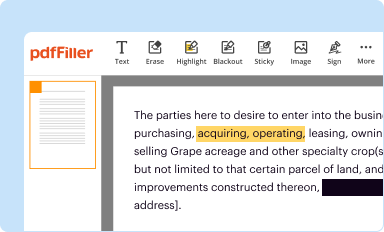
Edit your HHS FDA 3632 form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
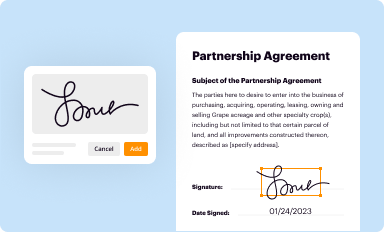
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your HHS FDA 3632 form via URL. You can also download, print, or export forms to your preferred cloud storage service.
Editing HHS FDA 3632 online
Use the instructions below to start using our professional PDF editor:
1
Set up an account. If you are a new user, click Start Free Trial and establish a profile.
2
Upload a document. Select Add New on your Dashboard and transfer a file into the system in one of the following ways: by uploading it from your device or importing from the cloud, web, or internal mail. Then, click Start editing.
3
Edit HHS FDA 3632. Rearrange and rotate pages, add new and changed texts, add new objects, and use other useful tools. When you're done, click Done. You can use the Documents tab to merge, split, lock, or unlock your files.
4
Get your file. Select the name of your file in the docs list and choose your preferred exporting method. You can download it as a PDF, save it in another format, send it by email, or transfer it to the cloud.
Dealing with documents is simple using pdfFiller.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
HHS FDA 3632 Form Versions
Version
Form Popularity
Fillable & printabley
How to fill out HHS FDA 3632
How to fill out HHS FDA 3632
01
Begin by downloading the HHS FDA 3632 form from the official FDA website.
02
Read the instructions carefully to understand the requirements.
03
Fill in the applicant's information, including name, address, and contact details.
04
Specify the purpose of the form in the designated section.
05
Provide detailed information related to the product or service being offered.
06
Include any necessary attachments or supporting documents as required.
07
Review all information for accuracy and completeness.
08
Sign and date the form where indicated.
09
Submit the form as per the instructions provided.
Who needs HHS FDA 3632?
01
Individuals or organizations applying for FDA approval or registration related to medical products.
02
Businesses involved in manufacturing or distributing regulated products.
03
Researchers needing FDA oversight for clinical studies.
04
Healthcare professionals involved in the development of FDA-regulated products.
Fill
form
: Try Risk Free






People Also Ask about
Why does the FDA regulate lasers?
The FDA may inspect manufacturers of laser products and require the recall of products that don't comply with federal standards or that have radiation safety defects. The agency also may test laser products and inspect displays of laser light shows to ensure the public is protected.
Which laser machine are FDA approved?
Candela™ GentleLASE®, Cynosure™ Apogee Elite® and Cutera™ CoolGlide® technologies are medical grade lasers that are manufactured in America and are FDA approved. Velvet Effect Lasers only use Candela™, Cynosure™ and Cutera™ technologies, not IPLs.
What is the FDA variance for lasers?
Under condition 13(a) of the variance application, beams must be kept at least 2.5 meters laterally and 3.0 meters vertically from audience access. This means laser beams should be kept approximately 10 feet above any surface upon which someone can stand, and approximately 8 feet in lateral separation.
What is the FDA registration?
Owners or operators of places of business (also called establishments or facilities) that are involved in the production and distribution of medical devices intended for use in the United States (U.S.) are required to register annually with the FDA. This process is known as establishment registration.
Where can I find FDA accession number?
Accession numbers are unique identifiers for reports in our database and are provided in the CDRH acknowledgement of receipt letters.
How do I get an FDA accession number?
How Do I Get an Accession Number for my Laser Product? In order to obtain an accession number, FDA requires manufacturers of most laser products to submit a product report consisting of numerous aspects including: Identifying information for the product and manufacturer.
Do I need an accession number?
Accession numbers are generally not required for referencing or citation purposes, the exception might be when there is insufficient bibliographic detail in a reference to identify it eg. NNN Museum.
What is an accession number for import?
Accession numbers are unique FDA identifiers for electronic product reports found in FDA's radiation-emitting electronic product database. FDA requires importers to provide the Accession Numbers on Form 2877. This is the one assigned by FDA to the manufacturers who made the imported electronic products.
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
Where do I find HHS FDA 3632?
The pdfFiller premium subscription gives you access to a large library of fillable forms (over 25 million fillable templates) that you can download, fill out, print, and sign. In the library, you'll have no problem discovering state-specific HHS FDA 3632 and other forms. Find the template you want and tweak it with powerful editing tools.
How do I edit HHS FDA 3632 straight from my smartphone?
Using pdfFiller's mobile-native applications for iOS and Android is the simplest method to edit documents on a mobile device. You may get them from the Apple App Store and Google Play, respectively. More information on the apps may be found here. Install the program and log in to begin editing HHS FDA 3632.
Can I edit HHS FDA 3632 on an iOS device?
No, you can't. With the pdfFiller app for iOS, you can edit, share, and sign HHS FDA 3632 right away. At the Apple Store, you can buy and install it in a matter of seconds. The app is free, but you will need to set up an account if you want to buy a subscription or start a free trial.
What is HHS FDA 3632?
HHS FDA 3632 is a form used by the U.S. Department of Health and Human Services and the Food and Drug Administration to gather information related to certain drug products.
Who is required to file HHS FDA 3632?
Manufacturers, repackagers, or relabelers of drugs that are subject to FDA regulation are required to file HHS FDA 3632.
How to fill out HHS FDA 3632?
HHS FDA 3632 should be filled out by providing relevant drug product information, including but not limited to drug names, usage, and manufacturing details, following the instructions provided by the FDA.
What is the purpose of HHS FDA 3632?
The purpose of HHS FDA 3632 is to ensure that the FDA has accurate and up-to-date information on drug products for regulatory oversight.
What information must be reported on HHS FDA 3632?
The information that must be reported includes product identification, manufacturing details, labeling information, and any other data required by the FDA regulations.
Fill out your HHS FDA 3632 online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

HHS FDA 3632 is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.
























