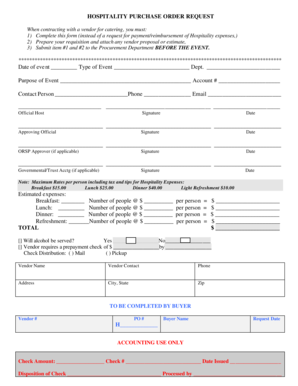
Get the free GLP Requirements Related to an Environment with NMR Systems User Guide. GLP Requirem...
Show details
GLP Requirements Related to an Environment with NMR Systems User Guide Version 004Innovation with IntegrityNMRCopyright by Bruker Corporation All rights reserved. No part of this publication may be
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign glp requirements related to

Edit your glp requirements related to form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.

Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your glp requirements related to form via URL. You can also download, print, or export forms to your preferred cloud storage service.
How to edit glp requirements related to online
Follow the steps below to use a professional PDF editor:
1
Register the account. Begin by clicking Start Free Trial and create a profile if you are a new user.
2
Prepare a file. Use the Add New button. Then upload your file to the system from your device, importing it from internal mail, the cloud, or by adding its URL.
3
Edit glp requirements related to. Text may be added and replaced, new objects can be included, pages can be rearranged, watermarks and page numbers can be added, and so on. When you're done editing, click Done and then go to the Documents tab to combine, divide, lock, or unlock the file.
4
Save your file. Select it from your records list. Then, click the right toolbar and select one of the various exporting options: save in numerous formats, download as PDF, email, or cloud.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out glp requirements related to

How to fill out glp requirements related to
01
Identify the specific GLP regulations applicable to your study or organization.
02
Gather all necessary documentation and protocols for your study.
03
Ensure that all personnel involved are trained in GLP compliant practices.
04
Establish a quality assurance program to monitor GLP compliance.
05
Document all experimental procedures accurately and thoroughly.
06
Implement a system for the proper storage and management of data and records.
07
Conduct regular audits and inspections to ensure ongoing compliance.
Who needs glp requirements related to?
01
Pharmaceutical companies conducting preclinical studies.
02
Biotechnology firms involved in research and development.
03
Laboratories performing non-clinical safety studies.
04
Academic institutions conducting research requiring regulatory compliance.
05
Organizations seeking to obtain regulatory approval for new products or treatments.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How can I manage my glp requirements related to directly from Gmail?
You can use pdfFiller’s add-on for Gmail in order to modify, fill out, and eSign your glp requirements related to along with other documents right in your inbox. Find pdfFiller for Gmail in Google Workspace Marketplace. Use time you spend on handling your documents and eSignatures for more important things.
How can I send glp requirements related to for eSignature?
When your glp requirements related to is finished, send it to recipients securely and gather eSignatures with pdfFiller. You may email, text, fax, mail, or notarize a PDF straight from your account. Create an account today to test it.
Can I sign the glp requirements related to electronically in Chrome?
You can. With pdfFiller, you get a strong e-signature solution built right into your Chrome browser. Using our addon, you may produce a legally enforceable eSignature by typing, sketching, or photographing it. Choose your preferred method and eSign in minutes.
What is glp requirements related to?
GLP (Good Laboratory Practice) requirements are related to the standards and guidelines that ensure the quality and integrity of laboratory data generated in non-clinical studies.
Who is required to file glp requirements related to?
Organizations and facilities that conduct non-clinical laboratory studies or research, typically including pharmaceutical companies, research institutions, and contract research organizations.
How to fill out glp requirements related to?
To fill out GLP requirements, laboratories must document their study protocols, maintain accurate records, and submit reports that detail testing methods, results, and compliance with GLP standards.
What is the purpose of glp requirements related to?
The purpose of GLP requirements is to ensure that non-clinical laboratory studies are conducted in a consistent, reproducible manner, safeguarding the quality of data and protecting human and environmental health.
What information must be reported on glp requirements related to?
Information that must be reported includes study objectives, methodologies, raw data, results, statistical analysis, and compliance with GLP standards.
Fill out your glp requirements related to online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Glp Requirements Related To is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.