
Get the free EPITOME-2: An open-label study assessing the ... - Pfizerpro.com.co
Show details
EPITOME2: An open label study assessing the transition to a new formulation of intravenous in patients with pulmonary arterial hypertension Olivier Simon, MD, a Marion Delacroix, MD, b Emmanuel Begot,
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign epitome-2 an open-label study
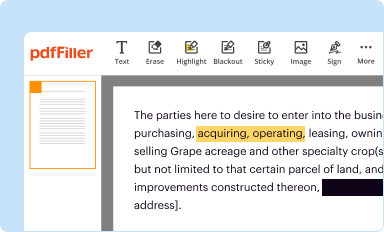
Edit your epitome-2 an open-label study form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
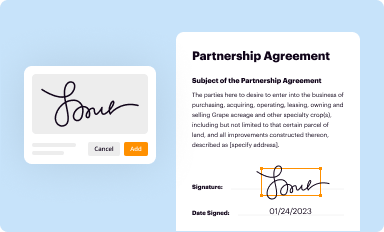
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your epitome-2 an open-label study form via URL. You can also download, print, or export forms to your preferred cloud storage service.
How to edit epitome-2 an open-label study online
Use the instructions below to start using our professional PDF editor:
1
Set up an account. If you are a new user, click Start Free Trial and establish a profile.
2
Simply add a document. Select Add New from your Dashboard and import a file into the system by uploading it from your device or importing it via the cloud, online, or internal mail. Then click Begin editing.
3
Edit epitome-2 an open-label study. Add and replace text, insert new objects, rearrange pages, add watermarks and page numbers, and more. Click Done when you are finished editing and go to the Documents tab to merge, split, lock or unlock the file.
4
Save your file. Select it from your list of records. Then, move your cursor to the right toolbar and choose one of the exporting options. You can save it in multiple formats, download it as a PDF, send it by email, or store it in the cloud, among other things.
It's easier to work with documents with pdfFiller than you can have believed. You can sign up for an account to see for yourself.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out epitome-2 an open-label study
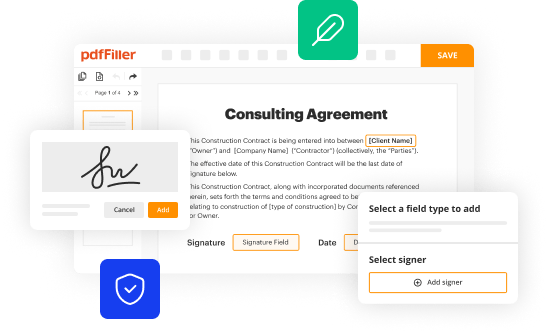
How to Fill Out Epitome-2 An Open-label Study:
01
Obtain the necessary study materials and documents: First, gather all the required forms, templates, and guidelines provided by the study sponsor. These materials will typically include the Epitome-2 study protocol, case report forms, informed consent forms, and any other relevant documentation.
02
Familiarize yourself with the study protocol: Thoroughly read and understand the study protocol provided. This document outlines the objectives, methodology, inclusion criteria, exclusion criteria, and other important details of the study. Make sure to comprehend the study design and procedures before proceeding further.
03
Collect patient data: Begin by collecting all necessary data from eligible patients who meet the inclusion criteria of the study. This may involve conducting interviews, physical examinations, medical tests, or reviewing medical records. Ensure that all data is accurately recorded and in compliance with ethical guidelines and patient privacy regulations.
04
Complete the case report forms (CRFs): Fill out the CRFs as per the instructions provided by the study sponsor. These forms are used to capture specific data points related to the study, such as demographic information, medical history, medication use, and any study-specific assessments. Double-check the information entered for accuracy and completeness.
05
Obtain informed consent: Before enrolling any patients into the study, obtain informed consent from them or their legal representatives. Ensure that the patients fully understand the study's purpose, procedures, potential risks, and benefits. Use the provided informed consent form, following all legal and ethical requirements.
06
Follow study procedures and guidelines: Adhere to the study procedures outlined in the protocol and any additional guidance provided. This may include administering study drugs, monitoring patient progress, conducting assessments, and documenting adverse events or side effects accurately.
07
Maintain proper documentation: Keep organized records of all study-related activities and documentation, including source data, consent forms, CRFs, and any correspondence with study personnel or participants. These records are crucial for ensuring data integrity, participant safety, and study compliance.
Who Needs Epitome-2 An Open-label Study:
01
Researchers: Scientists or investigators who want to study the effectiveness, safety, or other aspects of a specific treatment or intervention may require Epitome-2, an open-label study. By conducting such research, they aim to generate evidence and contribute to the scientific knowledge in their respective field.
02
Regulatory Bodies: Regulatory agencies such as the Food and Drug Administration (FDA) or the European Medicines Agency (EMA) may require an open-label study like Epitome-2 to assess the safety and effectiveness of a potential new drug or intervention before granting regulatory approval for its use in clinical practice.
03
Pharmaceutical Companies: Pharmaceutical companies often conduct open-label studies like Epitome-2 to gather additional data or confirm the findings of previous research on their drugs or interventions. This aids in developing marketing strategies, supporting regulatory submissions, and ensuring the safety and efficacy of their products.
04
Healthcare Providers: Healthcare providers, including doctors, nurses, and other healthcare professionals, may participate in Epitome-2 or similar open-label studies to gain access to new treatments or interventions for their patients. Participating in studies allows them to contribute to scientific knowledge and potentially offer their patients novel therapeutic options.
05
Patients: Patients who meet the study's inclusion criteria may enroll in Epitome-2 or similar open-label studies to access potentially beneficial treatments or interventions that are not yet widely available. By participating, patients contribute to scientific advancements and may have the opportunity to receive specialized care or investigational therapies.
Overall, Epitome-2, an open-label study, is valuable for researchers, regulatory bodies, pharmaceutical companies, healthcare providers, and patients to contribute to scientific knowledge, evaluate treatment effectiveness, and potentially gain access to innovative therapies.
Fill
form
: Try Risk Free






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How do I execute epitome-2 an open-label study online?
With pdfFiller, you may easily complete and sign epitome-2 an open-label study online. It lets you modify original PDF material, highlight, blackout, erase, and write text anywhere on a page, legally eSign your document, and do a lot more. Create a free account to handle professional papers online.
How do I edit epitome-2 an open-label study straight from my smartphone?
The best way to make changes to documents on a mobile device is to use pdfFiller's apps for iOS and Android. You may get them from the Apple Store and Google Play. Learn more about the apps here. To start editing epitome-2 an open-label study, you need to install and log in to the app.
Can I edit epitome-2 an open-label study on an Android device?
You can edit, sign, and distribute epitome-2 an open-label study on your mobile device from anywhere using the pdfFiller mobile app for Android; all you need is an internet connection. Download the app and begin streamlining your document workflow from anywhere.
What is epitome-2 an open-label study?
Epitome-2 is an open-label study is a type of research study where both the researchers and the participants know which treatment is being administered.
Who is required to file epitome-2 an open-label study?
Researchers conducting the study are required to file epitome-2 an open-label study.
How to fill out epitome-2 an open-label study?
The researchers must provide detailed information about the study design, participants, treatments, and outcomes in the epitome-2 form.
What is the purpose of epitome-2 an open-label study?
The purpose of epitome-2 open-label study is to evaluate the effectiveness and safety of a particular treatment.
What information must be reported on epitome-2 an open-label study?
Researchers must report detailed information about the study protocol, participant demographics, treatment administration, and study outcomes.
Fill out your epitome-2 an open-label study online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Epitome-2 An Open-Label Study is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.


















