Last updated on Oct 30, 2015

Get the free Commercial Invoice for Laboratory Samples
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Lab Commercial Invoice
The Commercial Invoice for Laboratory Samples is an invoice template used by researchers to document the shipping of non-commercial laboratory samples from the US to Taiwan.
pdfFiller scores top ratings on review platforms




Who needs Lab Commercial Invoice?
Explore how professionals across industries use pdfFiller.
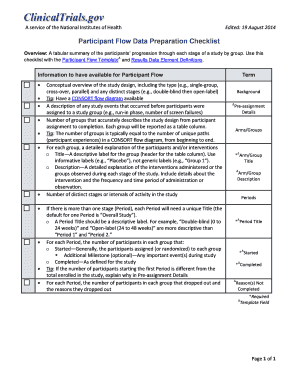
Comprehensive Guide to Lab Commercial Invoice
What is the Commercial Invoice for Laboratory Samples?
The commercial invoice for laboratory samples is a crucial document used during international shipping, specifically tailored for items designated for laboratory use. This invoice serves to declare the contents and their purpose, ensuring transparency in shipping laboratory samples from the U.S. to Taiwan. By using a precise commercial invoice template, shippers can aid customs officials in processing these items effectively.
Purpose and Benefits of the Commercial Invoice for Laboratory Samples
The importance of the commercial invoice lies in its ability to streamline customs clearance and confirm compliance with international shipping regulations. Utilizing a non-commercial goods invoice decreases the risk of delays or misunderstandings. The benefits extend to maintaining accurate records and facilitating efficient processing, which is essential for research institutions.
Key Features of the Commercial Invoice Template
A commercial invoice template typically includes several critical components essential for laboratory shipping. These components encompass:
-
Sender and recipient details
-
A comprehensive item description
-
Value, weight, and quantity of the samples
-
Clear declaration of the samples' non-commercial intent
-
Signature requirements, including those of the Research Director
The fillable nature of the form ensures that users can customize the fields according to the specifications of the shipment.
Who Needs the Commercial Invoice for Laboratory Samples?
The primary users of the commercial invoice for laboratory samples include research institutions and laboratories sending shipments internationally. The signature of a Research Director is necessary to validate the invoice, ensuring that the documentation meets regulatory standards.
How to Fill Out the Commercial Invoice for Laboratory Samples Online
To effectively complete the commercial invoice using pdfFiller, follow these steps:
-
Gather all required information, including sender and recipient details.
-
Describe each laboratory sample clearly, including its purpose.
-
Ensure that you include accurate values and weights.
-
Obtain the necessary signature from the Research Director.
-
Review all entries for accuracy before submission.
Field-by-Field Instructions for the Commercial Invoice
Each field in the commercial invoice is designed to capture specific information vital for the shipping process. Key fields include:
-
Sender's name and address
-
Recipient's name and contact information
-
Description of the laboratory samples
-
Value and total weight
-
Declaration of non-commercial intent
Common errors can be avoided by double-checking entries and confirming the accuracy of the Research Director's signature.
How to Sign the Commercial Invoice for Laboratory Samples
Signature requirements for the commercial invoice mandate the involvement of a Research Director. This adds a layer of credibility to the document. Users can choose between a digital signature and a wet signature, with each having its relevance depending on the submission method and regulations.
Where to Submit the Commercial Invoice for Laboratory Samples
Submission methods for the commercial invoice can vary, but typically include direct delivery to customs or shipping companies. It is advisable to track the submission status online or through automated systems provided by logistics partners to ensure timely processing.
Security and Compliance When Using the Commercial Invoice
Handling sensitive documents such as the commercial invoice necessitates a strong focus on data protection. pdfFiller employs multiple security features, including 256-bit encryption, ensuring compliance with HIPAA and GDPR standards for sensitive information.
Experience Hassle-Free Invoice Filling with pdfFiller
Utilizing pdfFiller for creating and managing the Commercial Invoice for Laboratory Samples simplifies the documentation process. With tools that enhance efficiency and security, users can ensure accurate and compliant form submissions with ease.
How to fill out the Lab Commercial Invoice
-
1.To access the Commercial Invoice for Laboratory Samples, navigate to pdfFiller's website and use the search bar to find the form by its name.
-
2.Once you have opened the form, you will see various fields that need to be completed, including sender and recipient information.
-
3.Before filling out the form, gather essential details such as the shipping address, description of laboratory samples, quantity, weight, and value of the goods.
-
4.Begin completing the form by filling in the sender’s name, address, and contact information in the appropriate fields.
-
5.Next, provide the recipient's information, making sure to double-check the address and contact details for accuracy.
-
6.Proceed to describe the goods being shipped, stating that the contents are intended for laboratory research and have no commercial value.
-
7.Input the necessary details concerning quantity, weight, and value of the samples to ensure compliance with shipping regulations.
-
8.If required, ensure that the Research Director's signature field is left blank for later completion, or sign it if you are authorized.
-
9.Once all fields are filled, review the entire document for any errors or missing information using the highlights from the pdfFiller interface.
-
10.After verifying the form, save your changes and download the completed invoice by clicking on the download button.
-
11.You may also submit the form directly through pdfFiller if that option is available, or print it for physical submission.

Who is eligible to use the Commercial Invoice for Laboratory Samples?
The form is intended for organizations and individuals involved in laboratory research who need to ship samples internationally, particularly from the US to Taiwan.
What is the importance of the Research Director's signature?
The Research Director's signature is required to validate the invoice, confirming that the samples are non-commercial and for laboratory research only.
Are there any deadlines for submitting this invoice?
While specific deadlines can vary by shipping provider, ensure that the invoice is completed and signed prior to the shipment date to prevent delays.
What supporting documents may be required with this invoice?
Depending on the shipping method, you may need to include additional documents such as export licenses, customs forms, or other regulatory paperwork.
What common mistakes should be avoided when filling out the form?
Ensure all fields are accurately completed, particularly recipient information and item descriptions, to avoid issues with customs or delivery.
How long does processing take for shipments with this invoice?
Processing times can vary based on the shipping method and destination. Typically, allow several days to a week for international shipments.
Can the Commercial Invoice be submitted electronically?
Yes, if your shipping service or customs authority accepts electronic documents, you can submit the invoice through pdfFiller or print it for physical submission.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.