Last updated on Dec 31, 2015

Get the free Clinical Trial Screening Enrollment Log
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Clinical Trial Log
The Clinical Trial Screening Enrollment Log is a healthcare document used by clinical trial coordinators to document and track potential subjects' eligibility in clinical trials.
pdfFiller scores top ratings on review platforms




Who needs Clinical Trial Log?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Clinical Trial Log
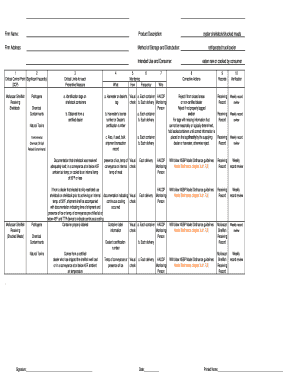
What is the Clinical Trial Screening Enrollment Log?
The Clinical Trial Screening Enrollment Log serves a crucial role in documenting potential subjects for clinical trials. This log is vital for maintaining an organized record, ensuring that all relevant information about each potential participant is captured systematically.
Key elements typically included in the clinical trial screening log are subject identifiers, screening dates, and tracking details that help streamline the recruitment process. By employing this log, researchers can effectively manage and monitor the candidate selection process throughout the trial.
Purpose and Benefits of the Clinical Trial Screening Enrollment Log
This clinical trial screening log offers several advantages in the research landscape. It facilitates strict adherence to standard operating procedures, ensuring that trials meet established regulatory and procedural standards.
Additionally, the log enhances tracking of subject eligibility and consent status, enabling researchers to make informed decisions based on accurate, up-to-date information. Utilizing an enrollment tracking form can significantly streamline the workflow associated with managing potential trial participants.
Key Features of the Clinical Trial Screening Enrollment Log
The clinical trial screening enrollment log is designed with several key features that enhance its usability. Fields within the log include consent status, eligibility criteria, and reasons for exclusion, which are essential for comprehensive record-keeping.
This fillable form integrates seamlessly into digital workflows, allowing researchers to document and access information efficiently. Such features ensure that the subject eligibility log remains a cornerstone of effective clinical trial documentation.
Who Needs the Clinical Trial Screening Enrollment Log?
The primary users of the clinical trial screening enrollment log include clinical trial coordinators and researchers who require an efficient method for tracking potential participants. These professionals are tasked with ensuring compliance with legal and ethical responsibilities surrounding human subjects in research.
Understanding the significance of a clinical trial consent form is crucial, as it directly impacts the ethical management of participants in clinical trials.
How to Fill Out the Clinical Trial Screening Enrollment Log Online (Step-by-Step)
Filling out the clinical trial screening enrollment log is a straightforward process that can be completed in several steps:
-
Open the log and enter subject identifiers in the designated fields.
-
Record the screening date and any necessary eligibility criteria.
-
Update the consent status and ensure all sections are completed accurately.
-
Review the information entered to avoid common pitfalls like incorrect data.
Taking care to accurately record information will enhance the usability of the enrollment tracking form throughout the trial.
Review and Validation Checklist for the Clinical Trial Screening Enrollment Log
To ensure the clinical trial screening enrollment log is complete and accurate before submission, consider the following checklist:
-
Confirm all fields are completed thoroughly.
-
Verify the accuracy of entered data, especially consent and eligibility information.
-
Encourage peer reviews or validations from colleagues or supervisors to minimize errors.
How to Save and Share the Clinical Trial Screening Enrollment Log
Saving and sharing the completed clinical trial screening enrollment log can be done effectively by following these best practices:
-
Utilize document formats that are easy to download, such as PDF.
-
Consider cloud storage options for secure access and management.
-
Explore solutions for securely sharing the log with team members or regulatory bodies to maintain compliance.
Security and Compliance Considerations
Handling the clinical trial screening enrollment log necessitates a strong focus on security and compliance. It is essential to remain compliant with regulations like HIPAA, ensuring that data protection measures are in place.
pdfFiller prioritizes secure handling of sensitive documents, employing 256-bit encryption to protect user information and maintain privacy and data protection standards.
Common Errors and How to Avoid Them
Several common errors may arise when using the clinical trial screening enrollment log. Users often experience challenges such as missing information or inaccuracies in data entry. These mistakes can significantly impact clinical trial processes.
To mitigate errors, it is advisable to double-check all entered information and remain vigilant during the data entry process to ensure the documentation represents accurate records of potential subjects.
Get Started with pdfFiller for Your Clinical Trial Screening Enrollment Log
To enhance your form-filling experience, consider using pdfFiller for your clinical trial screening enrollment log. This platform offers an intuitive interface, security features, and the ability to create fillable forms that streamline clinical documentation tasks.
By leveraging pdfFiller, you can efficiently manage your clinical trial records, ensuring a smoother workflow and improved organizational efficiency during trial operations.
How to fill out the Clinical Trial Log
-
1.Access the Clinical Trial Screening Enrollment Log on pdfFiller by searching for the document title in the platform's search bar.
-
2.Once you locate the form, click on it to open and load the document for editing.
-
3.Before starting, gather necessary information about potential subjects, including identifiers, screening dates, and consent statuses.
-
4.Navigate through the form using pdfFiller's user-friendly interface, filling in fields such as subject identifiers with the corresponding data.
-
5.Use checkboxes provided in the form to mark consent status and eligibility criteria for each subject.
-
6.For any additional notes or reasons for exclusion, fill in the designated text fields clearly and concisely.
-
7.Once you have completed all required fields, review the entered information for accuracy and completeness.
-
8.Make sure there are no errors or missing information before finalizing the form.
-
9.To finalize the form, look for the save option provided in pdfFiller to keep a record of your completed document.
-
10.You can also download the form directly to your device or submit it as required through pdfFiller's options.

What are the eligibility requirements for participants?
Eligibility requirements typically vary based on clinical trial protocols. Generally, potential subjects must meet specific criteria related to age, health conditions, and other factors outlined in the trial's inclusion/exclusion criteria.
Is there a deadline for completing the enrollment log?
While the enrollment log should be filled out as potential subjects are screened, it is important to submit it in tandem with trial deadlines set by the study protocol or regulatory requirements.
How do I submit the completed log?
Completed logs can be submitted through the designated submission channels outlined in the clinical trial protocol. Ensure all logs are filled accurately before submission to avoid delays.
What supporting documents are required with the enrollment log?
Typically, you may need to include supporting documents such as informed consent forms and participant medical history, as they confirm eligibility and consent for participation.
What common mistakes should I avoid when filling in the log?
Common mistakes include entering incorrect subject identifiers, neglecting to update consent statuses, and leaving required fields blank. Always review the log for completeness before submission.
What is the processing time for the enrollment log?
Processing time may vary according to the regulatory requirements of the trial. However, logs should be reviewed promptly to ensure that potential subjects can be enrolled without delay.
Are there specific concerns about handling sensitive information in the log?
Yes, it is critical to handle sensitive participant information with confidentiality. Follow HIPAA guidelines to ensure that all personal health information is protected while managing the enrollment log.
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.