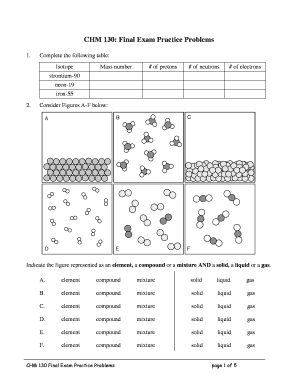
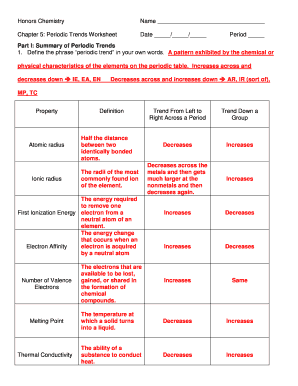
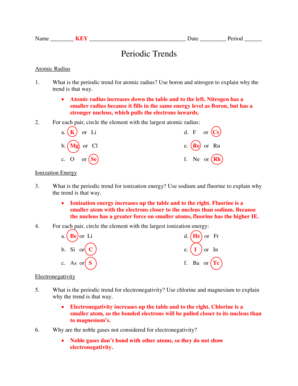
Electronegativity Chart
What is Electronegativity Chart?
An electronegativity chart is a tool used in chemistry to determine the relative ability of an atom to attract electrons in a chemical bond. It provides a numerical value, known as electronegativity, which helps in understanding the polarity of a molecule and predicting its behavior in chemical reactions. The electronegativity chart ranks elements based on their electronegativity values, with fluorine having the highest electronegativity of 4.0.
What are the types of Electronegativity Chart?
There are mainly two types of electronegativity charts used in chemistry:
How to complete Electronegativity Chart
Completing an electronegativity chart involves the following steps:
Remember that while electronegativity values provide useful information about the behavior of elements in chemical bonds, they are not the only factor determining bond strength and polarity. Other factors such as bond length and molecular geometry also play important roles. As an SEO writer, I strongly recommend using pdfFiller, an online document editing platform that empowers users to create, edit, and share documents effortlessly. With unlimited fillable templates and powerful editing tools, pdfFiller is the perfect PDF editor you need to get your documents done efficiently.