
Get the free retention form
Show details
Form-300 Issue date: Finished Goods Retention Sample Register Log (Ref: SOP LAB-045) Date started: Date completed: The following are details of Retention Samples contained in the box: Product Code
We are not affiliated with any brand or entity on this form
Get, Create, Make and Sign retention form
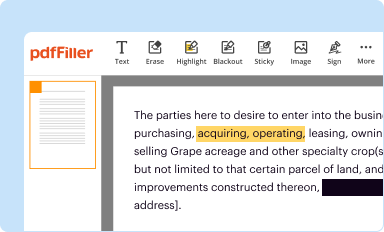
Edit your retention form form online
Type text, complete fillable fields, insert images, highlight or blackout data for discretion, add comments, and more.
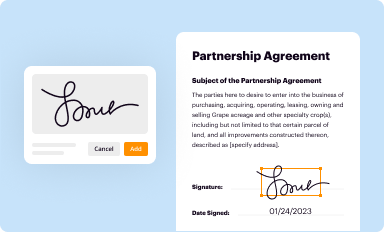
Add your legally-binding signature
Draw or type your signature, upload a signature image, or capture it with your digital camera.

Share your form instantly
Email, fax, or share your retention form form via URL. You can also download, print, or export forms to your preferred cloud storage service.
How to edit retention form online
Here are the steps you need to follow to get started with our professional PDF editor:
1
Register the account. Begin by clicking Start Free Trial and create a profile if you are a new user.
2
Prepare a file. Use the Add New button to start a new project. Then, using your device, upload your file to the system by importing it from internal mail, the cloud, or adding its URL.
3
Edit retention form. Rearrange and rotate pages, add and edit text, and use additional tools. To save changes and return to your Dashboard, click Done. The Documents tab allows you to merge, divide, lock, or unlock files.
4
Save your file. Select it from your list of records. Then, move your cursor to the right toolbar and choose one of the exporting options. You can save it in multiple formats, download it as a PDF, send it by email, or store it in the cloud, among other things.
With pdfFiller, it's always easy to deal with documents.
Uncompromising security for your PDF editing and eSignature needs
Your private information is safe with pdfFiller. We employ end-to-end encryption, secure cloud storage, and advanced access control to protect your documents and maintain regulatory compliance.
How to fill out retention form
How to fill out form-300 finished goods retention:
01
Start by gathering all the required information and documents. This may include the product description, quantity, date of retention, and any supporting documents related to the retention.
02
Next, carefully read the instructions provided with the form. Make sure you understand the purpose of the form and the information required in each section.
03
Begin filling out the form by entering your personal information and contact details. This may include your name, address, phone number, and email.
04
Move on to the section where you need to provide details about the finished goods retention. Fill in the required information such as the name of the product, quantity retained, date of retention, and the reason for retention.
05
If there are any additional details or remarks related to the retention, make sure to include them in the designated section.
06
Review the filled-out form carefully to ensure all the information is accurate and complete.
07
If required, sign and date the form to certify the information provided.
Who needs form-300 finished goods retention:
01
Companies or businesses that have a need to retain finished goods for a certain period of time.
02
Organizations that need to keep track of inventory or maintain records of stored goods.
03
Industries that require proper documentation and traceability of retained finished goods for regulatory or compliance purposes.
Note: It is important to consult with relevant authorities or legal professionals to determine if your specific situation requires the use of form-300 finished goods retention.
Fill
form
: Try Risk Free






People Also Ask about
What is a control sample?
Control samples are reference materials with a matrix composition close to that of the analyzed samples, and which elemental mass fractions are close to the expected ones in the unknown samples. They should also be stable over time, and be available in sufficient amount as to be used for years.
How do you store retention samples?
Retention samples should be kept at the testing facility where the study was conducted. The study sponsor should provide the testing facility with a supply of the test article and the reference standard sufficient to complete the study and retain the appropriate number of dosage units as reserve samples.
What is the retention period of samples?
Reference and retention samples from each batch of finished product should be retained for at least one year after the expiry date.
What is a retention sample of a finished product?
Retention sample: a sample of a fully packaged unit from a batch of finished product. It is stored for identification purposes. For example, presentation, packaging, labelling, patient information leaflet, batch number, expiry date should the need arise during the shelf life of the batch concerned.
What is the difference between control sample and retention sample?
An appropriately identified sample that is representative of each batch that shall be retained is known as control sample. These are also referred as retention or reserve sample. Control samples shall be collected for Finished Product, Raw Material and Packing Materials.
What is a retention sample?
A reference sample is a sample for the purpose of future analysis, which could refer to starting materials, packaging materials or finished products. A retention sample is a sample representing the batch of finished product as distributed. Samples from a stability trial program cannot be used as retention samples.
For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How do I edit retention form in Chrome?
Get and add pdfFiller Google Chrome Extension to your browser to edit, fill out and eSign your retention form, which you can open in the editor directly from a Google search page in just one click. Execute your fillable documents from any internet-connected device without leaving Chrome.
How can I edit retention form on a smartphone?
You can do so easily with pdfFiller’s applications for iOS and Android devices, which can be found at the Apple Store and Google Play Store, respectively. Alternatively, you can get the app on our web page: https://edit-pdf-ios-android.pdffiller.com/. Install the application, log in, and start editing retention form right away.
How do I edit retention form on an Android device?
You can edit, sign, and distribute retention form on your mobile device from anywhere using the pdfFiller mobile app for Android; all you need is an internet connection. Download the app and begin streamlining your document workflow from anywhere.
What is form-300 finished goods retention?
Form-300 finished goods retention is a document used to report and retain records regarding finished goods inventory for compliance and regulatory purposes.
Who is required to file form-300 finished goods retention?
Manufacturers and suppliers of finished goods who meet specific regulatory requirements are typically required to file form-300 finished goods retention.
How to fill out form-300 finished goods retention?
To fill out form-300 finished goods retention, entities must provide details such as the description of the goods, quantities held, and relevant identifiers, while ensuring accuracy and compliance with guidelines.
What is the purpose of form-300 finished goods retention?
The purpose of form-300 finished goods retention is to ensure accurate tracking and reporting of finished goods inventory, aiding in regulatory compliance and inventory management.
What information must be reported on form-300 finished goods retention?
Information reported on form-300 finished goods retention typically includes product descriptions, inventory quantities, production dates, and location of goods.
Fill out your retention form online with pdfFiller!
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

Retention Form is not the form you're looking for?Search for another form here.
Relevant keywords
Related Forms
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.
























