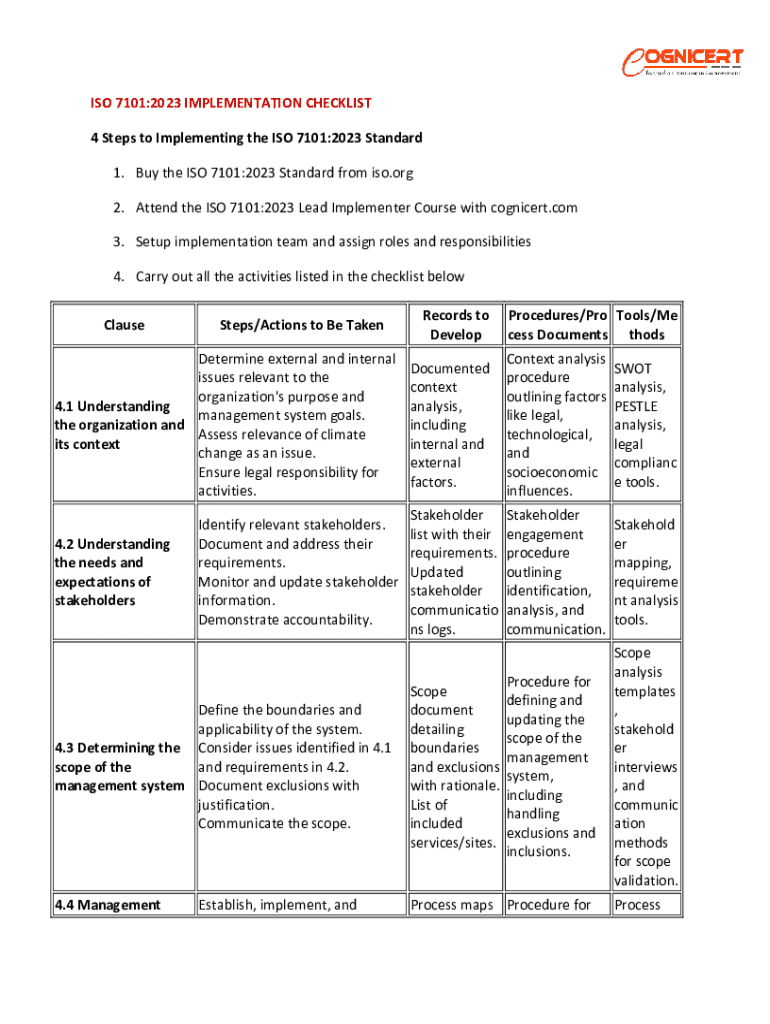
Get the free ISO 7101 Implementation Guide
Get, Create, Make and Sign iso 7101 implementation guide

Editing iso 7101 implementation guide online
Uncompromising security for your PDF editing and eSignature needs
How to fill out iso 7101 implementation guide
How to fill out iso 7101 implementation guide
Who needs iso 7101 implementation guide?
ISO 7101 Implementation Guide Form: A Comprehensive How-To Guide
Overview of ISO 7101 Standards
ISO 7101 refers to a set of international standards established to ensure effective management and measurement for various processes in organizations. These standards are crucial for maintaining efficiency, improving quality, and enhancing patient safety in sectors such as healthcare, manufacturing, and service industries. Compliance with ISO 7101 enables organizations to benchmark their operations against recognized global best practices, fostering trust among stakeholders, including employees, customers, and regulatory bodies.
The importance of compliance with ISO 7101 standards cannot be overstated. Achieving these standards not only boosts an organization's reputation but also paves the way for continuous improvement in quality management practices. Furthermore, ISO 7101 serves as a foundation for organizations striving for healthcare excellence by establishing frameworks that prioritize patient safety and operational compliance.
ISO 7101 broadly applies across various industries, including healthcare organizations that utilize the standards to standardize protocol, enhance patient care processes, and ensure regulatory compliance. In manufacturing, these standards optimize production processes, thus minimizing waste and inefficiencies. The versatility of ISO 7101 demonstrates its integral role in fostering quality culture across multiple sectors.
Understanding the ISO 7101 Implementation Guide Form
The ISO 7101 Implementation Guide Form is a practical tool designed to help organizations navigate the complexities of implementing the ISO 7101 standards. This form consolidates critical information and processes involved in achieving compliance and serves as a roadmap for systematic action. By using this guide, organizations can easily track their progress and ensure that all necessary steps are completed efficiently.
Key elements included in the ISO 7101 Implementation Guide Form typically consist of baseline information such as organizational structure, compliance measures, responsible parties, and timelines for implementation. By structuring the document this way, the form becomes a powerful facilitator of compliance, as it helps organizations to methodically define and document their pathways to meeting ISO 7101 standards.
This implementation guide is not only a compliance tool but also a mechanism for continuous improvement, encouraging teams to reflect on existing processes and identify areas for refinement. Thus, utilizing the ISO 7101 Implementation Guide Form leads to not only adherence to standards but also fosters a culture of quality and efficiency within the organization.
Who needs the ISO 7101 Implementation Guide Form?
The ISO 7101 Implementation Guide Form is crucial for several target audiences, primarily organizations seeking ISO 7101 certification. These entities require structured documentation to navigate the certification process efficiently. By using the form, they can outline their compliance commitment clearly and create a structured approach to fulfilling ISO standards.
In addition, professionals engaged in quality management play a pivotal role in utilizing this form. They are responsible for ensuring that the implementation process aligns with the relevant standards, continuously monitoring progress, and maintaining documentation. Teams tasked with compliance documentation also find the form indispensable, as it simplifies the gathering, recording, and reporting of essential information.
Step-by-step guide to completing the ISO 7101 Implementation Guide Form
Step 1: Gather necessary documentation
Before filling out the ISO 7101 Implementation Guide Form, it is essential to gather all necessary documentation. This includes existing policies and procedures, compliance records, employee training materials, and any prior audit reports. Having these documents readily available not only streamlines the process but ensures that all information is accurate and comprehensive.
Step 2: Fill out the base information
The next step is to enter the base information on the form. This includes your organization's name, address, and contact information, as well as the relevant ISO 7101 certification details. Accuracy in this section is critical to ensure that all future correspondence regarding compliance and certification is correctly routed and recorded.
Step 3: Outline compliance measures
Articulating compliance measures is essential in demonstrating how your organization intends to adhere to ISO 7101 standards. Detail each procedure and protocol you have in place to achieve compliance. Common compliance measures might include quality management systems, tracking and reporting protocols, and continuous improvement plans.
Step 4: Identify responsible parties
It is vital to identify who within your organization will be responsible for implementing each aspect of the compliance measures. Clearly defining roles prevents confusion and ensures accountability throughout the implementation process. Best practices dictate that roles align with individuals' expertise and current responsibilities.
Step 5: Establish a timeline for implementation
Creating a realistic timeline is key to keeping the implementation process on track. When establishing deadlines, factor in resource availability, workload distribution, and the complexity of compliance measures to set achievable goals. A well-defined timeline provides motivation and clarity as your organization progresses towards compliance.
Step 6: Review and finalize the form
Once all sections have been filled out, reviewing entries for completeness is crucial. Engage stakeholders to gather feedback, ensuring that all aspects have been considered. This is especially critical in compliance documentation, as collective insight can unearth potential oversights before submission.
Tips for effective use of the ISO 7101 Implementation Guide Form
Utilizing the ISO 7101 Implementation Guide Form effectively requires a structured approach. Firstly, ensure that you remain organized, maintaining clarity in documentation that avoids unnecessary confusion. Transitioning into compliance should enhance efficiency; thus, integrating digital tools like pdfFiller can facilitate real-time collaboration. Ensure that various team members can contribute and edit documents from different locations to streamline the process.
Moreover, be vigilant about common pitfalls such as neglecting to keep records current or underestimating the time required for implementation. Frequent error-checking can prevent issues from escalating, reinforcing a culture of improvement. Adopting best practices, such as regular progress meetings and shared responsibilities, cultivates team trust and commitment to achieving ISO compliance.
Frequently asked questions about the ISO 7101 Implementation Guide Form
Many questions arise concerning the utilization of the ISO 7101 Implementation Guide Form. Common inquiries include how to succinctly capture compliance measures and what level of detail is necessary. Organizations often wonder about the best ways to update the form after submission and how to maintain an ongoing compliance culture after receiving certification.
It's vital to approach these challenges with a proactive mindset. Seeking input from team members and industry experts can enhance your strategy and troubleshooting capabilities. Engaging in continuous education regarding ISO standards and compliance helps organizations stay ahead of regulatory changes and fosters a sustainable success model.
Benefits of using pdfFiller for ISO 7101 documentation
pdfFiller provides unparalleled support for organizations looking to manage ISO 7101 documentation effectively. Its seamless editing and collaboration features allow multiple users to contribute to a document concurrently, enhancing workflow efficiency. The cloud-based access empowers teams located in different geographic regions to collaborate in real-time, improving communication and reducing delays in response times.
In addition to great teamwork capabilities, pdfFiller’s eSigning features promote quick approval processes, enabling documents to be executed without unnecessary delays. Moreover, by providing ongoing document management solutions, pdfFiller ensures that organizations can maintain control over their documentation long after initial compliance efforts, solidifying their commitment to quality improvement.
Support and training resources
When utilizing the ISO 7101 Implementation Guide Form, access to support and training resources is essential. pdfFiller offers a comprehensive suite of resources to assist users through every stage of the documentation process. From detailed tutorials to live customer support, organizations can find assistance tailored to their needs, ensuring they fully understand how to leverage the form for maximum effectiveness.
Many organizations also find value in scheduled training sessions focusing on the specific aspects of ISO 7101 compliance. Such sessions foster a deeper understanding of how to align internal processes with the standards, enhancing overall operational effectiveness. By investing in training and support, companies position themselves strategically for sustained success.
Real-life success stories
Organizations leveraging the ISO 7101 Implementation Guide Form have seen remarkable improvements in compliance and operational performance. For instance, a mid-sized healthcare organization was able to streamline its documentation process, significantly improving patient safety outcomes through better quality monitoring. These success stories often highlight not just compliance achievements but also tangible benefits like enhanced trust from patients and the community.
Testimonials from teams using pdfFiller consistently emphasize advantages such as ease of use and collaboration efficiency, leading to quicker turnaround times for compliance documentation. The stories indicate that the combination of a structured approach to ISO 7101 and innovative tools results in a strong culture of quality that supports continuous improvement.
Next steps for your ISO 7101 compliance journey
After completing the ISO 7101 Implementation Guide Form, organizations should focus on next steps for ongoing compliance management. Establishing a regular audit schedule to review processes and update documentation reinforces a long-term commitment to ISO standards. Furthermore, consider setting up a feedback loop that allows team members to voice suggestions for continuous improvement, ensuring engagement remains high.
Additional resources, including industry seminars and updates on best practices, can provide valuable insights for sustaining compliance. By fostering a proactive approach, organizations will not only benefit from the certification but also improve operational dynamics in alignment with quality initiatives critical for healthcare excellence.






For pdfFiller’s FAQs
Below is a list of the most common customer questions. If you can’t find an answer to your question, please don’t hesitate to reach out to us.
How can I manage my iso 7101 implementation guide directly from Gmail?
How do I make changes in iso 7101 implementation guide?
How do I complete iso 7101 implementation guide on an iOS device?
What is iso 7101 implementation guide?
Who is required to file iso 7101 implementation guide?
How to fill out iso 7101 implementation guide?
What is the purpose of iso 7101 implementation guide?
What information must be reported on iso 7101 implementation guide?
pdfFiller is an end-to-end solution for managing, creating, and editing documents and forms in the cloud. Save time and hassle by preparing your tax forms online.

























