How to create a Medical Device Validation Report Template with pdfFiller
The Medical Device Validation Report Template Generator Tool by pdfFiller allows users to effortlessly create, edit, and manage validation reports for medical devices in PDF format. This tool is particularly beneficial for teams seeking a streamlined and accessible solution for document creation and collaboration.
What is a Medical Device Validation Report Template?
A Medical Device Validation Report Template is a structured document used to outline the validation process for medical devices, ensuring compliance with regulatory standards and providing detailed assessments of device functionality, safety, and efficacy. This template helps organizations follow best practices in device validation, streamlining the process and facilitating consistency across reports.
Why you might need a Medical Device Validation Report Template?
Organizations involved in medical device development and manufacturing often require these templates for several reasons:
-
1.To ensure compliance with regulatory standards set by agencies such as the FDA and EMA.
-
2.To maintain accurate records of validation processes, which can be vital during audits.
-
3.To provide a clear format that captures essential information succinctly.
-
4.To enhance collaboration across teams by using a standard template.
Key tools in pdfFiller that let you create Medical Device Validation Report Templates
pdfFiller offers a range of tools to assist users in generating Medical Device Validation Report Templates efficiently:
-
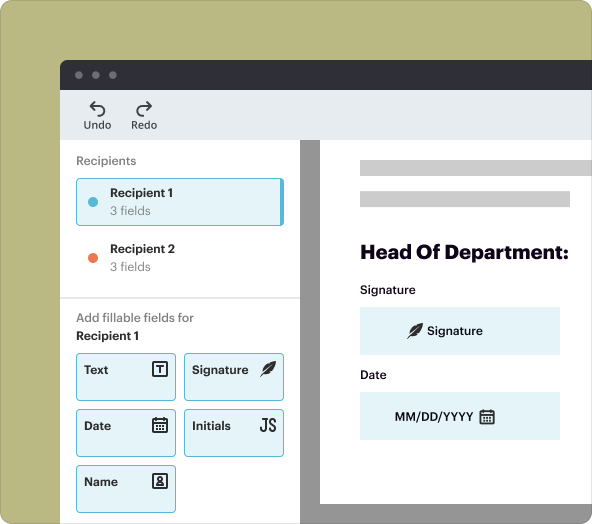
1.Document Editor: A comprehensive tool for editing text, images, and other elements of PDF files.
-
2.eSignature: Enable secure signing of documents electronically, facilitating timely approvals.
-
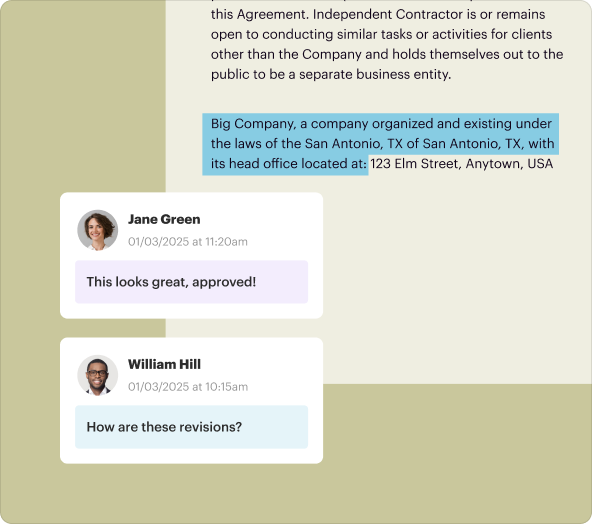
3.Collaboration Tools: Share documents with team members for real-time collaboration and revisions.
-
4.Cloud Storage: Access templates and completed reports from anywhere, ensuring flexibility.
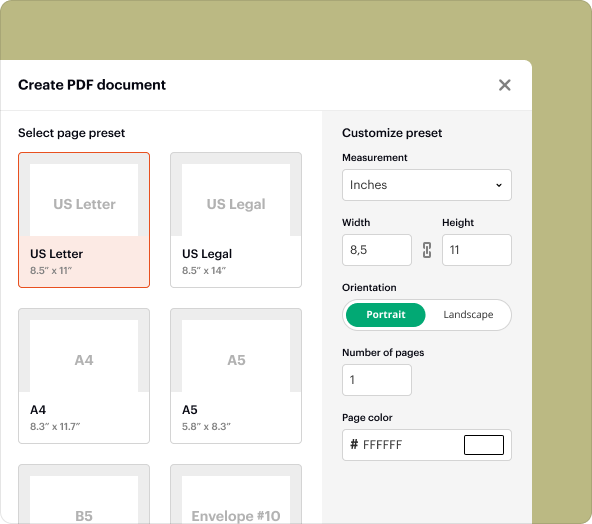
Step-by-step guide to creating Medical Device Validation Report Template blank PDFs
Generating a blank Medical Device Validation Report Template in pdfFiller is straightforward. Follow these steps:
-
1.Log into your pdfFiller account.
-
2.Select the ‘Create New Document’ option from the dashboard.
-
3.Choose ‘Blank Document’ as your starting point.
-
4.Add form fields as necessary, such as text fields and checkboxes.
-
5.Customize the layout and formatting to meet your specific needs.
Creating Medical Device Validation Report Templates from scratch vs. uploading existing files to modify
Depending on your requirements, you can either create a Medical Device Validation Report Template from scratch or modify an existing document. Here are the pros and cons of each method:
From Scratch
-
1.Pros: Complete control over layout, elements, and customization.
-
2.Cons: Time-consuming and may require more effort to ensure all necessary sections are included.
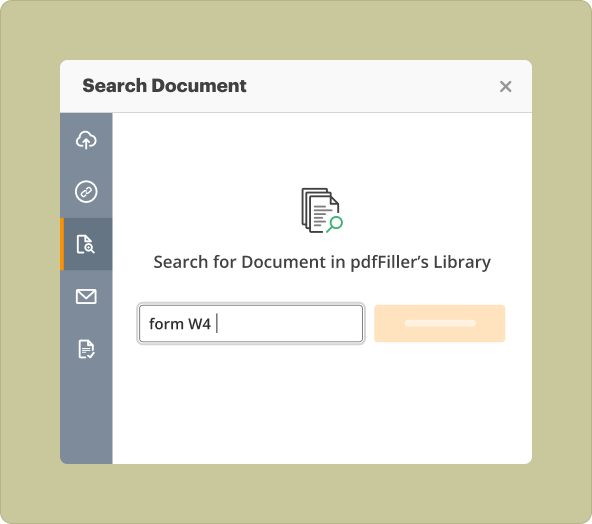
Uploading Existing Files
-
1.Pros: Faster process if starting with a partially completed report; easily modifies existing content.
-
2.Cons: Limited by the structure of the original document; may require extensive editing.
Organizing content and formatting text as you create Medical Device Validation Report Templates
Effective organization and formatting can enhance the clarity of your validation report. Utilize the following tips in pdfFiller:
-
1.Use headers and footers to include important details, like document titles and versions.
-
2.Position text fields logically to guide the user through the report.
-
3.Incorporate bullet points and lists to break down information into digestible chunks.
-
4.Utilize tables for data presentation, ensuring readability and accessibility.
Saving, exporting, and sharing once you create a Medical Device Validation Report Template
After creating your Medical Device Validation Report Template, pdfFiller provides several options for saving, exporting, and sharing your document:
-
1.Saving: Save your document in the cloud for easy access and future editing.
-
2.Exporting: Download the document in various formats, including PDF, Word, and more.
-
3.Sharing: Share the document via email or generate a link for collaborators to access.
Typical use-cases and sectors that often utilize Medical Device Validation Report Templates
Several industries depend on precise Medical Device Validation Report Templates, including:
-
1.Healthcare: Hospitals and clinics requiring compliance documentation for medical devices.
-
2.Manufacturing: Companies involved in the production and quality assurance of medical equipment.
-
3.Regulatory Affairs: Professionals managing submissions and audits with regulatory bodies.
-
4.Research: Institutions documenting clinical trials and equipment effectiveness.
Conclusion
The Medical Device Validation Report Template Generator Tool from pdfFiller empowers individuals and teams to create, manage, and collaborate on essential documentation efficiently. By utilizing its comprehensive set of features, users can ensure compliance, enhance collaboration, and streamline their document creation process. Whether starting from scratch or modifying existing templates, pdfFiller's intuitive platform meets diverse documentation needs across various sectors.
How to create a PDF with pdfFiller
Who needs this?
Document creation is just the beginning
Manage documents in one place
Sign and request signatures
Maintain security and compliance
pdfFiller scores top ratings on review platforms