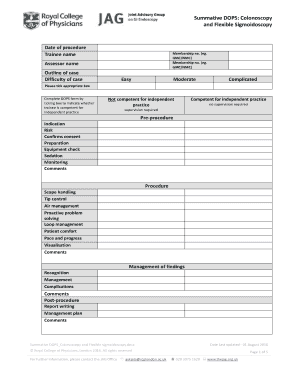
WPS PQR Qualified Range free printable template
We are not affiliated with any brand or entity on this form
Why choose pdfFiller for your legal forms?
All-in-one solution
pdfFiller offers a PDF editor, eSignatures, file sharing, collaboration tools, and secure storage—all in one place.
Easy to use
pdfFiller is simple, cloud-based, has a mobile app, and requires no downloads or a steep learning curve.
Secure and compliant
With encryption, user authentication, and certifications like HIPAA, SOC 2 Type II, and PCI DSS, pdfFiller keeps sensitive legal forms secure.
pdfFiller scores top ratings on review platforms




So far, looks like what I been looking for.
Easy to use -- several format/file extension options for saving and ability to link with cloud and sharing platforms makes this so versatile. Having the option to save a filled form as a template is very helpful. The customization and array of options is allowing me to complete tasks, for which I previously needed multiple programs.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.