Okay Signatory Clinical Trial Agreement Template For Free




Users trust to manage documents on pdfFiller platform
Send documents for eSignature with signNow
Watch a quick video tutorial on how to Okay Signatory Clinical Trial Agreement Template
pdfFiller scores top ratings in multiple categories on G2
Okay Signatory Clinical Trial Agreement Template in minutes
pdfFiller allows you to Okay Signatory Clinical Trial Agreement Template in no time. The editor's hassle-free drag and drop interface allows for fast and user-friendly document execution on any operaring system.
Ceritfying PDFs electronically is a fast and secure way to validate documents anytime and anywhere, even while on the fly.
See the detailed guide on how to Okay Signatory Clinical Trial Agreement Template electronically with pdfFiller:
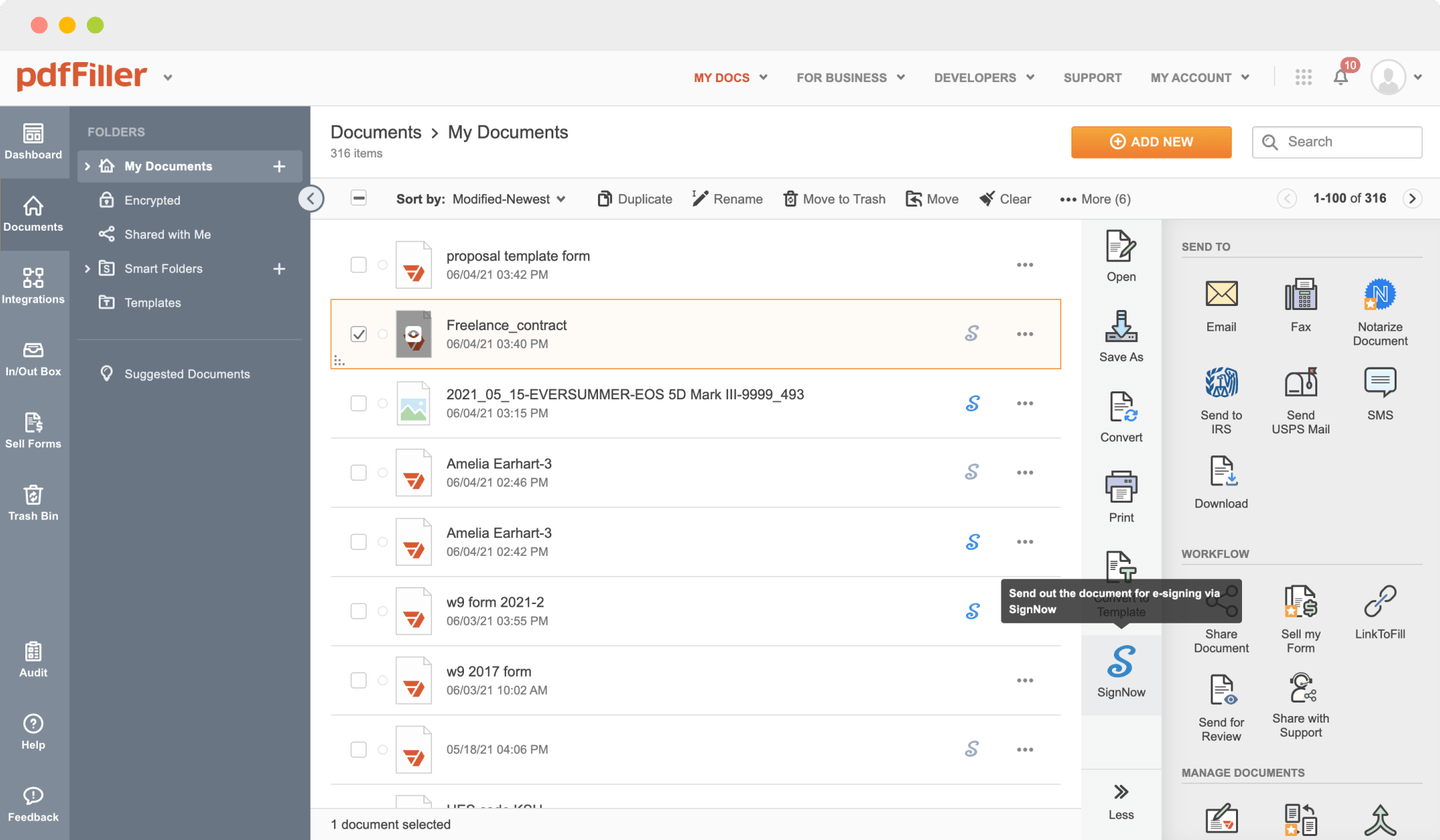
Add the document for eSignature to pdfFiller from your device or cloud storage.
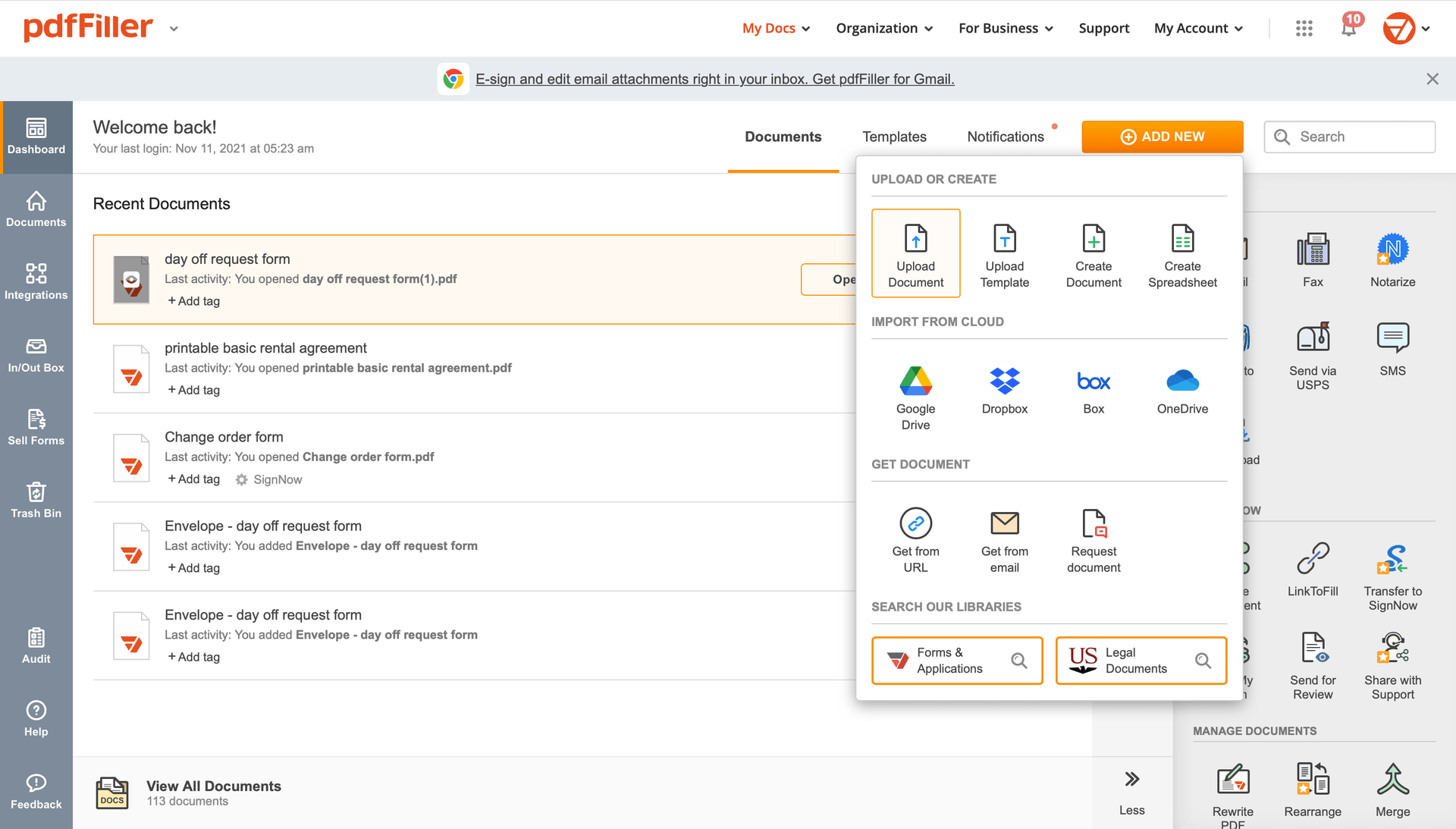
Once the file opens in the editor, hit Sign in the top toolbar.

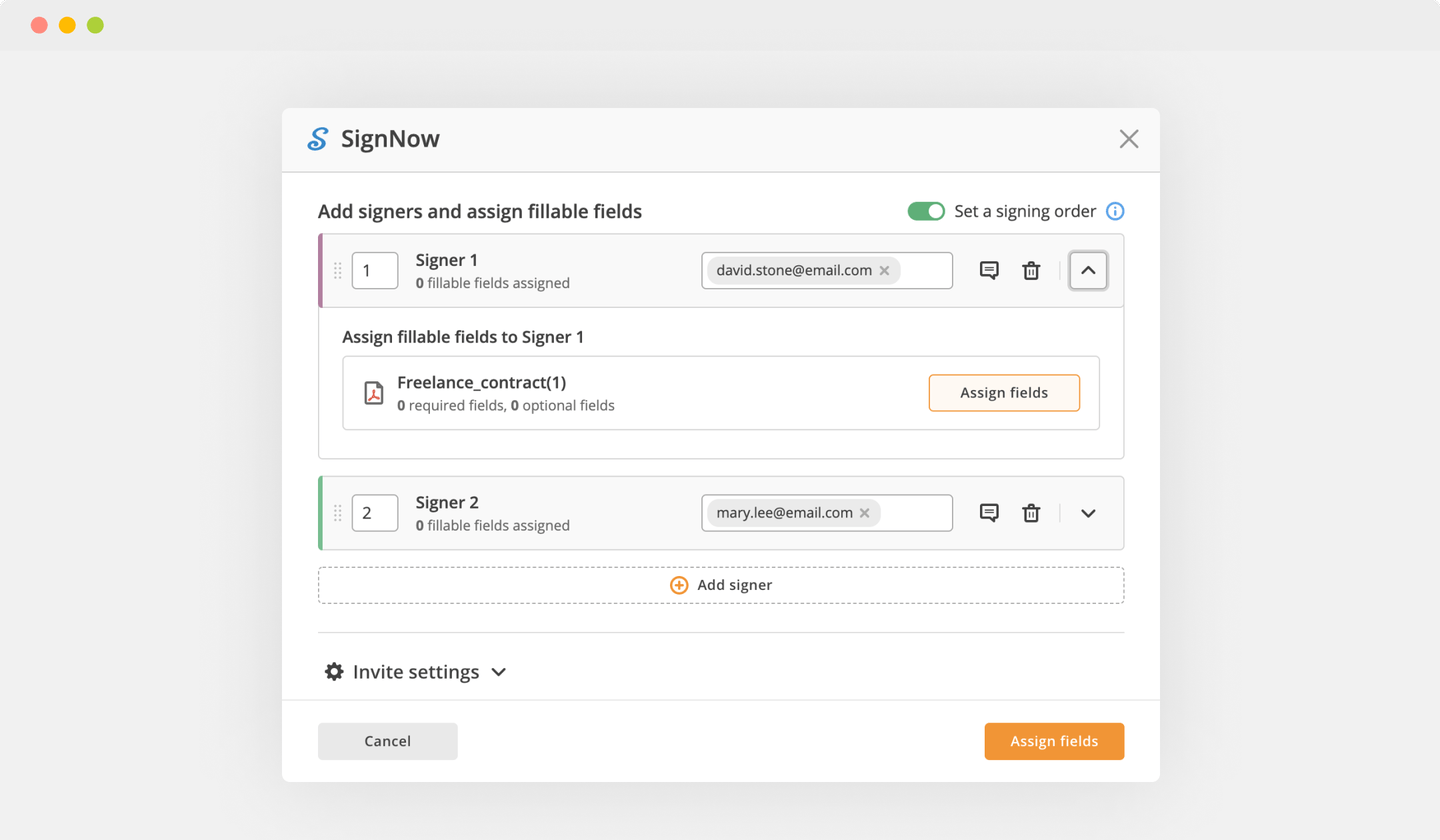
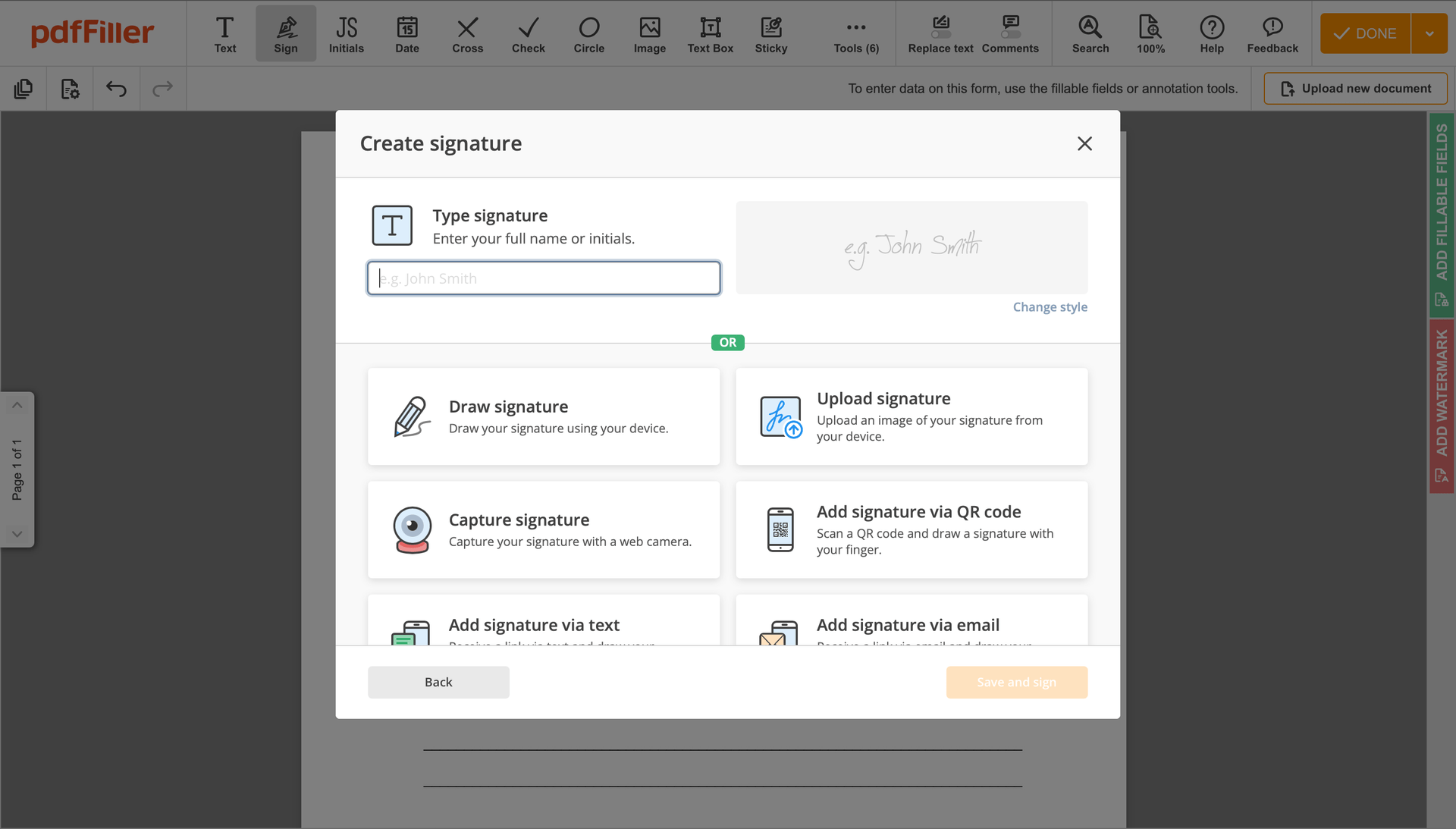
Create your electronic signature by typing, drawing, or importing your handwritten signature's image from your laptop. Then, click Save and sign.
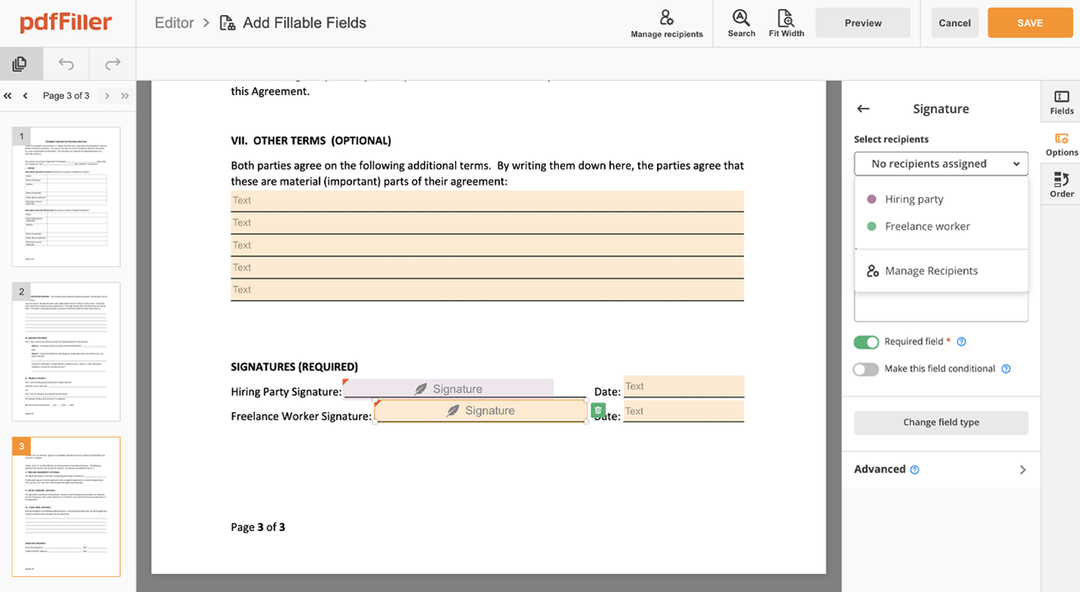
Click anywhere on a form to Okay Signatory Clinical Trial Agreement Template. You can move it around or resize it using the controls in the hovering panel. To use your signature, hit OK.
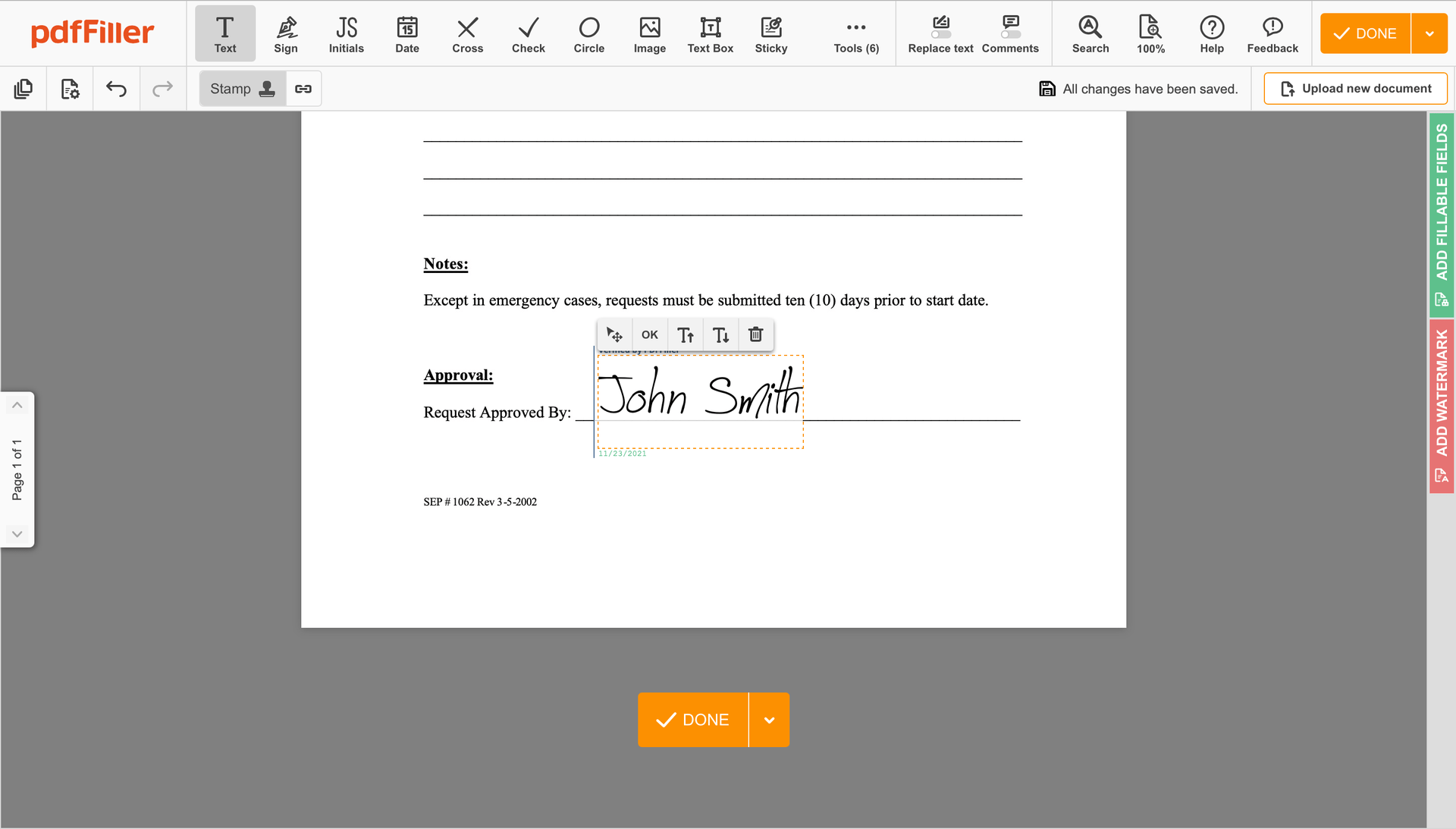
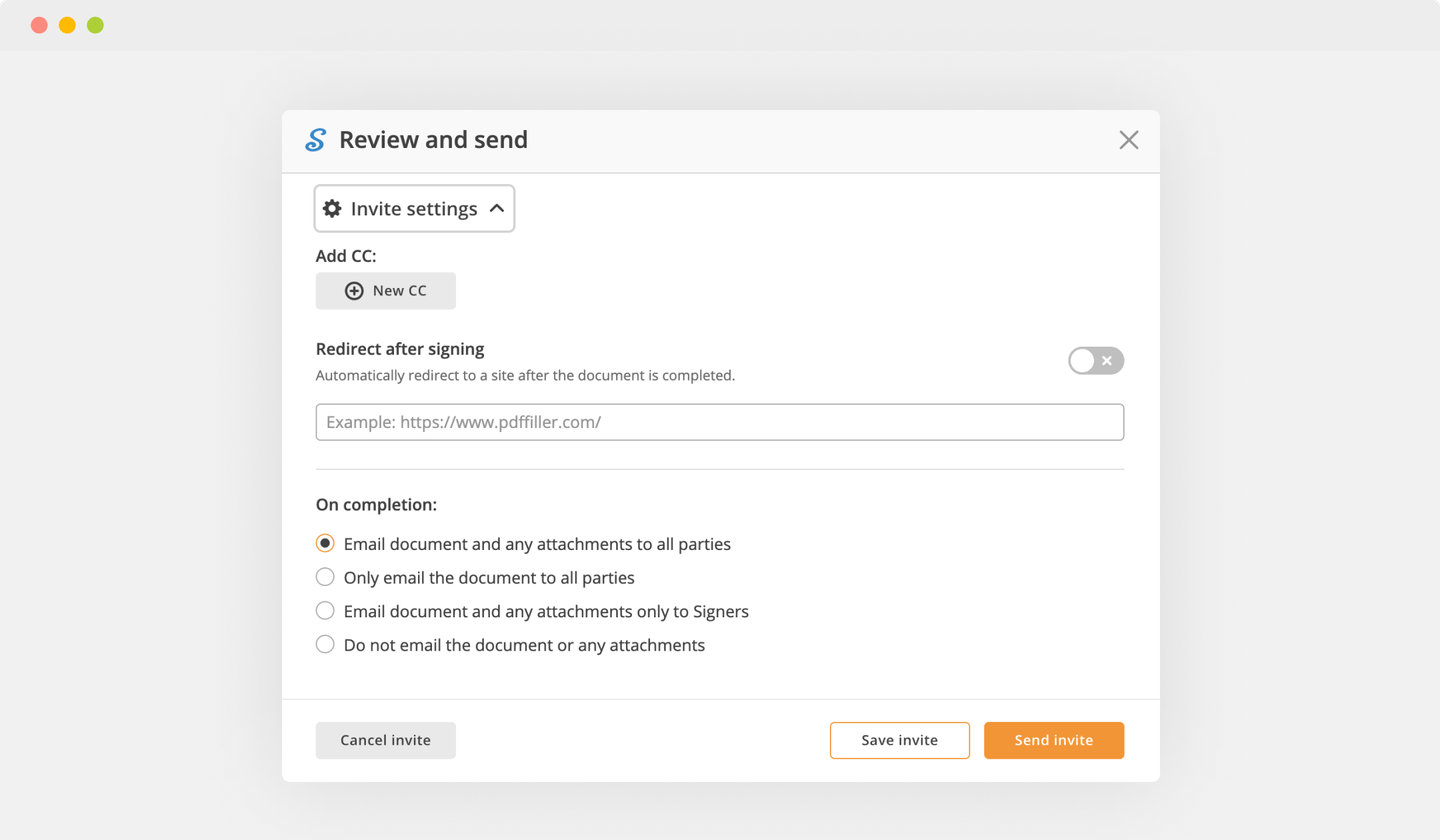
Finish up the signing session by hitting DONE below your form or in the top right corner.
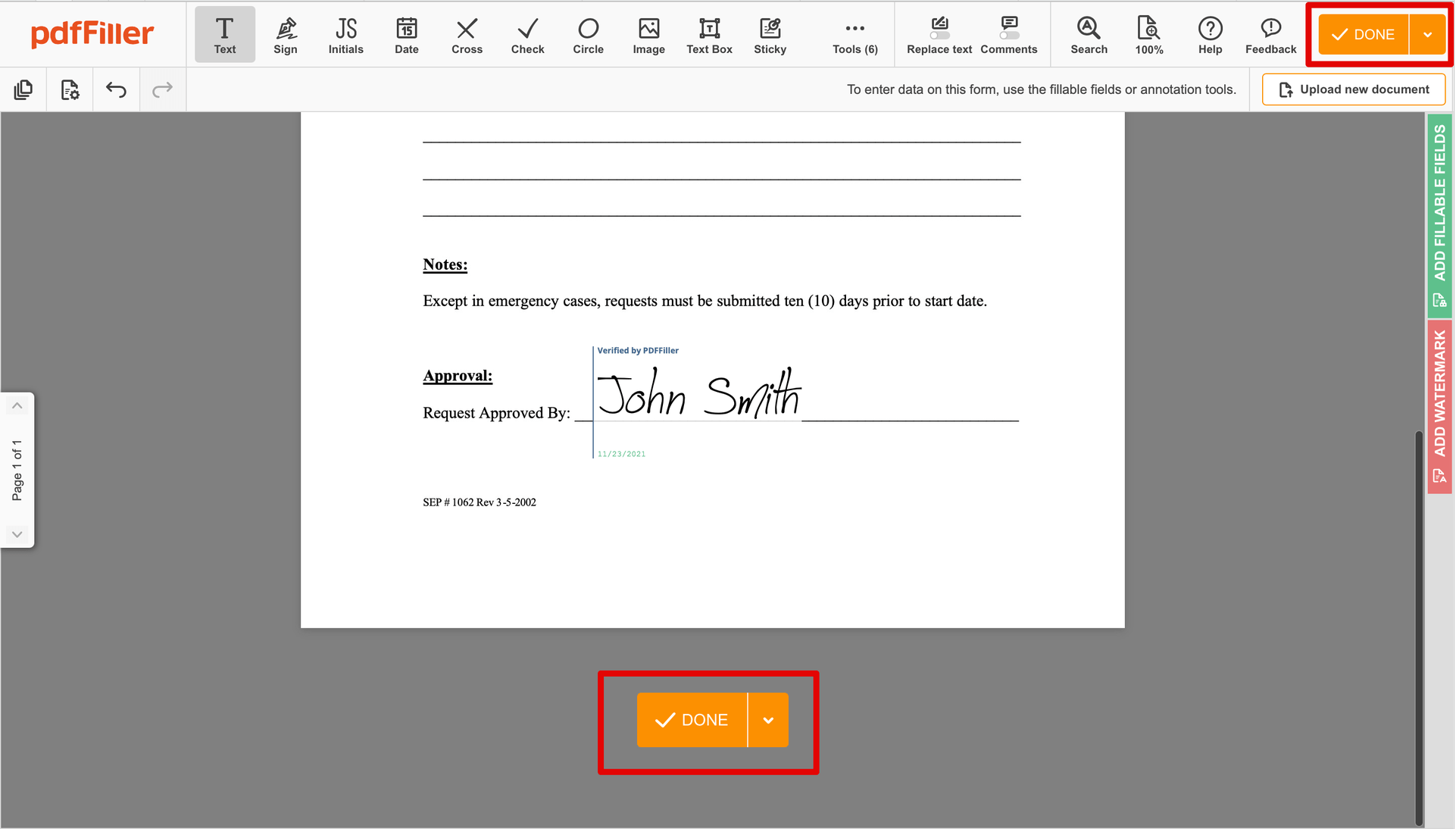
After that, you'll go back to the pdfFiller dashboard. From there, you can download a signed copy, print the form, or send it to other parties for review or validation.
Still using multiple applications to manage and edit your documents? Try this solution instead. Use our tool to make the process efficient. Create forms, contracts, make templates, integrate cloud services and utilize other features without leaving your account. Plus, the opportunity to Okay Signatory Clinical Trial Agreement Template and add high-quality features like signing orders, alerts, requests, easier than ever. Get the value of full featured platform, for the cost of a lightweight basic app.
How to edit a PDF document using the pdfFiller editor:
How to Send a PDF for eSignature
What our customers say about pdfFiller